
a)
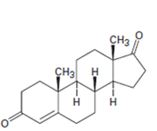
Interpretation:
Starting from testosterone how to prepare the compound shown is to be shown.
Concept introduction:
Pyridiniumchlorochromate in dichloromethane oxidizes 10 alcohols to
To show:
Starting from testosterone how to prepare the compound shown is to be stated.
b)
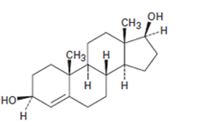
Interpretation:
Starting from testosterone how to prepare the compound shown is to be stated.
Concept introduction:
LiAlH4 in ether reduces unsaturated aldehydes, acids and esters to 10 alcohols. It reduces ketones to 20 alcohols. The double bond remains unaffected during the reduction.
To state:
Starting from testosterone how to prepare the compound shown.
c)
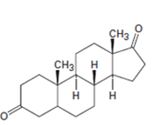
Interpretation:
Starting from testosterone how to prepare the compound shown is to be stated.
Concept introduction:
Pyridiniumchlorochromate in dichloromethane oxidizes 10 alcohols to aldehydes and 20 alcohols to ketones. H2, Pd/C can reduce the double bond in a compound without affecting aldehydic or keto group present in the compound.
To state:
Starting from testosterone how to prepare the compound shown.
d)
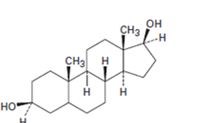
Interpretation:
Starting from testosterone how to prepare the compound shown is to be stated.
Concept introduction:
LiAlH4 in ether reduces unsaturated aldehydes, acids and esters to 10 alcohols. It reduces ketones to 20 alcohols. The double bond remains unaffected during the reduction. The double bond can be reduced using H2, Pd/C.
To state:
Starting from testosterone how to prepare the compound shown.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Organic Chemistry
- One frequently used method for preparing methyl esters is by reaction of carboxylic acids with diazomethane, CH2N2. The reaction occurs in two steps: (l) protonation of diazomethane by the carboxylic acid to yield methyldiazonium ion, CH3N2+, plus a carboxylate ion; and (2) reaction of the carboxylate ion with CH3N2+. (a) Draw two resonance structures of diazomethane, and account for step 1. (b) What kind of reaction occurs in step 2?arrow_forwardFormaldehyde, H2C=O, is known to all biologists because of its usefulness as a tissue preservative. When pure, formaldehyde trimerizes to give trioxane, C3H6O3, which, surprisingly enough, has no carbonyl groups. Only one monobromo derivative (C3H5BrO3) of trioxane is possible. Propose a structure for trioxane.arrow_forward13-21 The reaction of bromine with toluene in the presence of FeCl3 gives a mixture of three products, all with the molecular formula C7H7Br. Name and draw a structural formula for each product.arrow_forward
- 13-29 Show that if you add Steps 2a and 2b of the radical- chain mechanism for the autoxidation of a fatty acid hydrocarbon chain, you arrive at the following net equation: H I —CH2CH=CH—CH— + 0—0 • » Section of a fatty acid Oxygen hydrocarbon chain O—O—H I —CH2CH=CH—CH— A hydroperoxidearrow_forwardAldehydes and ketones react with thiols to yield thioacetals just as they react with alcohols to yield acetals. Predict the product of the following reaction, and propose a mechanism:arrow_forwardIdentify (A) in the following reaction. 2H2 Pt (A) KMNO4 Warm conc. || С — С — о—н |CO,H + HO CO2H cis-cyclo hexane 1,2-dicarboxylic acid (a) (b) (c) (d)arrow_forward
- Triethanolamine, (HOCH2CH2)3N, is a widely used biological buffer, with maximum buffering capacity at pH 7.8. Propose a synthesis of this compound from ethylene oxide and ammonia.arrow_forward(a) Explain how NaBH, in CH;OH can reduce hemiacetal A to 1,4-butanediol (HOCH,CH,CH,CH,OH). (b) What product is formed when A is treated with Ph;P=CHCH,CH(CH),? (c) The drug isotretinoin is formed by reaction of X and Y. What is the structure of isotretinoin? Although isotretinoin (trade name Accutane or Roaccutane) is used for the treatment of severe acne, it is dispensed under strict controls because it also causes birth defects. PPha NaOCH,CH3 HO- isotretinoin HO A Br X Yarrow_forwardPredict the products of the following acid-base reactions. If the equilibrium would not result in the formation of appreciable amounts of products, you should so indicate. In each case label the stronger acid, the stronger base, the weaker acid, and the weaker base: (a) CH3CH=CH2 + NANH2 (d) CH3C=C: + CH;CH2OH → (e) CH3C=C:- + NH¾CI – | (b) CH;C=CH + NaNH2 (c) CH3CH2CH3 + NANH2 → | HASarrow_forward
- (b) Predict the suitable solvent (H2O or CH3COCH3) to increase the reaction of bromopropane (CH3CH2CH2B1) with sodium hydroxide (NaOH). Two reactions are shown below: NaOH, 55 °C CH;CH,CH,Br CH;CH,CH,OH + NaBr H,O (i) NaOH, 55 °C CH;CH,CH,Br CH;CH,CH,OH NaBr H,C CH (ii)arrow_forwardWhich of the following reactions will synthesize phenol from benzene? 1) HNO3 + H2SO4; 2) Fe, HCl; 3) NaNO2, HCl, 0-10 oC; 4) warm H2SO4 and H2O 1) HNO3 + H2SO4; 2) Fe, HCl; 3) NaNO2, HCl, 0-10 oC; 4) CuCN; 5) dilute acid and heat 1) Acetyl chloride & AlCl3; 2) bleach 1) Ph-N2+ + KI; 2) BrMgCH=CH2 in ether, followed by H3O+; 3) warm, conc'd KMnO4 1) Cl-CH(CH3)-CH2CH2CH3 + FeBr3; 2) hot, conc'd KMnO4arrow_forwardProvide the major product for the following reaction? (1) BH3, ether (2) H2O2, OHarrow_forward

 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning




