
Concept explainers
a)
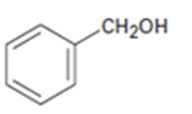
Interpretation:
All possible compounds which give benzyl alcohol on reduction with LiAlH4 are to be given.
Concept introduction:
All carbonyl compounds, including acids, esters,
To give:
All possible compounds which give benzyl alcohol on reduction with LiAlH4.
b)
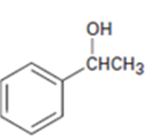
Interpretation:
All possible compounds which give 1-phenylethanol on reduction with LiAlH4 are to be given.
Concept introduction:
All carbonyl compounds, including acids, esters, aldehydes and ketones are reduced by LiAlH4. Acids, esters and aldehydes yield a 10 alcohol upon reduction with LiAlH4, while ketones yield 20 alcohols. The reaction is carried out in ether solution as LiAlH4 reacts violently with water and explodes when heated above 120oC.
To give:
All possible compounds which give 1-phenylethanol on reduction with LiAlH4.
c)
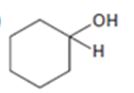
Interpretation:
All possible compounds which give cyclohexanol on reduction with LiAlH4 are to be given.
Concept introduction:
All carbonyl compounds, including acids, esters, aldehydes and ketones are reduced by LiAlH4. Acids, esters and aldehydes yield a 10 alcohol upon reduction with LiAlH4, while ketones yield 20 alcohols. The reaction is carried out in ether solution as LiAlH4 reacts violently with water and explodes when heated above 120oC.
To give:
All possible compounds which give cyclohexanol on reduction with LiAlH4.
d)
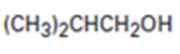
Interpretation:
All possible compounds which give isobutyl alcohol on reduction with LiAlH4 are to be given.
Concept introduction:
All carbonyl compounds, including acids, esters, aldehydes and ketones are reduced by LiAlH4. Acids, esters and aldehydes yield a 10 alcohol upon reduction with LiAlH4, while ketones yield 20 alcohols. The reaction is carried out in ether solution as LiAlH4 reacts violently with water and explodes when heated above 120oC.
To give:
All possible compounds which give isobutyl alcohol on reduction with LiAlH4.
Trending nowThis is a popular solution!

Chapter 17 Solutions
Organic Chemistry
- Treatment of cis-4-bromocyclohexanol with HO− affords compound Aand cyclohex-3-en-1-ol. Treatment of trans-4-bromocyclohexanol under the same conditions forms compound B and cyclohex-3-en-1-ol. A and Bcontain different functional groups and are not isomers of each other.Propose structures for A and B and offer an explanation for theirformation.arrow_forwardAcid-catalyzed hydrolysis of the following epoxide gives a trans diol. Of the two possible trans diols, only one is formed. How do you account for this stereoselectivity?arrow_forwardDraw the skeletal structures of all the final organic products formed when phenol (C6H-OH) is treated with the set of reagents given below: OH HNO₂ H₂SO4 1) Sn, HC1 2) dilute aq NaOH ?arrow_forward
- Devise a synthesis of the ketone hexan-3-one, CH3CH2COCH2CH2CH3, from CH3CH2Br as the only organic starting material; that is, all the carbon atoms in hexan-3-one must come from CH3CH2Br. You may use any other neededreagents.arrow_forwardSynthesize the following compounds from 4 carbon or less mono-functional alcohols, benzene, toluene and any organic or inorganic reagents needed, NH2arrow_forwardWhich one of the following compounds gives 5-methyl-3-heptanol with LiAlH4 followed by water?arrow_forward
- Starting from Cyclohexanol as the only organic reagent you have and use any other inorganic reagents to prepare:arrow_forwardPrepare the following compounds : 1-Starting with benzene or any other reagents , prepare 3-phenyl -1-propanolarrow_forwardPropose a synthesis of the topical anesthetic cyclomethycaine from 4-hydroxybenzoic acid, 2-methylpiperidine, and any other necessary reagents. HN' + HO. НО Cyclomethycaine 4-Hydroxybenzoic acid 2-Methylpiperidinearrow_forward
- The following diol is prepared from propanal (CH3CH2CHO) and 3-bromopropanol. Which derivatives will be among intermediate products in this transformation? ОН HOarrow_forwardShow how cyclohexanecarboxylic acid could be synthesized from cyclohexane in good yield. -COOHarrow_forwardQII: Starting from toluene as the only organic reagent you have and use any other inorganic reagents to prepare: ОН -CH НС CH,arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

