
Concept explainers
a)
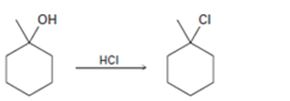
Interpretation:
The complete mechanism for the conversion of 30 alcohol given to 30
Concept introduction:
Alcohols get protonated in the presence of strong acids. The protonated intermediate will lose a molecule of water to yield a carbocation. The nucleophilic attack by the halide ion will lead to the formation of the alkyl halide.
To propose:
The complete mechanism for the conversion of 30 alcohol given to 30 alkyl halide by treating with HCl involving two cationic intermediates.
b)
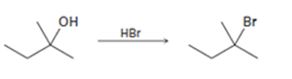
Interpretation:
The complete mechanism for the conversion of 30 alcohol given to 30 alkyl halide by treating with HBr involving two cationic intermediates is to be proposed.
Concept introduction:
Alcohols get protonated in the presence of strong acids. The protonated intermediate will lose a molecule of water to yield a carbocation. The nucleophilic attack by the halide ion will lead to the formation of the alkyl halide.
To propose:
The complete mechanism for the conversion of 30 alcohol given to 30 alkyl halide by treating with HBr involving two cationic intermediates.
c)
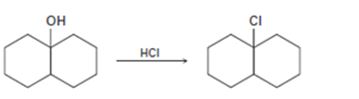
Interpretation:
The complete mechanism for the conversion of 30 alcohol given to 30 alkyl halide by treating with HCl involving two cationic intermediates is to be proposed.
Concept introduction:
Alcohols get protonated in the presence of strong acids. The protonated intermediate will lose a molecule of water to yield a carbocation. The nucleophilic attack by the halide ion will lead to the formation of the alkyl halide.
To propose:
The complete mechanism for the conversion of 30 alcohol given to 30 alkyl halide by treating with HCl involving two cationic intermediates.
Trending nowThis is a popular solution!

Chapter 17 Solutions
Organic Chemistry
- The conversion of 3 alcohols into alkenes under acidic conditions involves two cationic intermediates. For each reaction, draw the complete mechanism using curved arrows.arrow_forwardDraw out the complete mechanism of the reaction and include all relevant electrons and arrows showing electron movement. NaOHarrow_forwardQ- mahesh Please draw out the mechanism and explain the reagents you use.arrow_forward
- 4. Draw the mechanism for the following reaction using curved arrows. i NaBH4 CH3OH OHarrow_forwardDraw a mechanism for the following reaction. OH H+ + H₂Oarrow_forward5) Use a curved arrow notation mechanism to show how each product is formed in the following reaction. Iderstity-themejor prodret. 0H HzO Br OHarrow_forward
- Draw out a complete arrow-pushing mechanism for the following reaction. This reaction is explicitly done with heat. O NaOH, H₂O heatarrow_forwardComplete the following stepwise reaction mechanism problems based on the reaction conditions given: Draw the stepwise mechanism.arrow_forwardDraw the reaction mechanism for the reaction between an aldehyde and water under acidic conditions. (H') H,0arrow_forward
- Draw a step-wise mechanism for the following substitution reaction. Be sure to add lone pairs and charges where relevant. CI H₂Oarrow_forwardAddition of water to an alkyne gives a keto‑enol tautomer product. Draw the ketone that is in equilibrium with the given enol.arrow_forwardDraw the most stable product formed in each of the reactions shown. Reaction (a) ich NaOEt, EtOH Draw the product of reaction (a). Rings Select Draw H Morearrow_forward
