
Concept explainers
Interpretation: Among below molecules, molecules that are capable of hydrogen bonding to them should be determined.
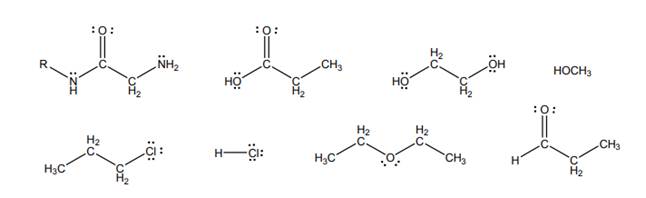
Concept introduction: Intermolecular forces are types of forces that are responsible to hold atoms together in molecule. There are various types of intermolecular forces as follows:
1. Hydrogen bonding:
As is evident from its name, these types of forces exist if hydrogen is present. Such forces are present when hydrogen atom bonds with highly electronegative elements like
2. Ion-dipole forces:
This force exists between ion and molecules with dipole moment in them. Attraction exists between ion and oppositely charged end of dipole.
3. Ion-induced dipole forces:
When an ion approaches non-polar molecule, it induces temporary dipole in it. Ion gets attracted towards oppositely charged part of dipole induced in molecule.
4. Dispersion forces:
These forces are also known as induced dipole-induced dipole or van der Waals forces. Such forces are present between various atoms and molecules. These are observed generally in non-polar molecules, halogens, and noble gases.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Organic Chemistry: A Guided Inquiry
- The boiling point of the following compounds is expected to decrease in this order (highest boiling point first).arrow_forwardRank molecules A-D (1=lowest, 4=highest) in order of increasing boiling point. Briefly rationalize your ranking. Please answer all parts.arrow_forwardLabel the kind of intermolecular interaction that will dominate in each of the following molecules.arrow_forward
- I find it pretty hard to understand the difference in boiling points when just given the shape of the molecule. Please help!arrow_forwardExplain the difference between the nature of the bonds between water molecules and those found between ethyl alcohol molecules, and how that plays a role in what you observed.arrow_forwardCircle all of the following that exhibit ONLY dispersion attractions between molecules. Explain your choices. A) XeF4 B)AsH3 C)CO2 D) CF4 E)H2 F)CHCl3arrow_forward
- Select the diatomic molecule below that has the highest boiling point. * F₂ Cl₂ Br₂ I₂arrow_forwardWhich of these molecules exhibit hydrogen bonding? H. H. H -C-H H-C-CI H C -OH H. H. H. methane chloromethane methanol Select all that apply. methanol chloromethane methanearrow_forwardOrder the molecules by increasing low to high boiling pointsarrow_forward
- Which is neither polar nor capable of hydrogen bonding? Explain with example?arrow_forwardWhich of the following sets of atoms will NOT form a strong hydrogen bond? (Explain your answer) a. C-H .. 0-C b. 0-H .. N ....... c. N-H d. 0-H ....... .......arrow_forwardOn the vitamin c molecule in a 3d structure position the arrow blue dashed arrows so that it points to a region where London forces are likely to be operating and position the pink arrow so that it points to a region where hydrogen bonding is likely to be operating.arrow_forward
 Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

