
Concept explainers
The following pictures represent initial concentrations in solutions that contain a weak acid HA (
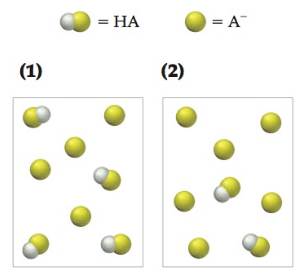
(a) Which solution has the highest pH? Which has the lowest pH?
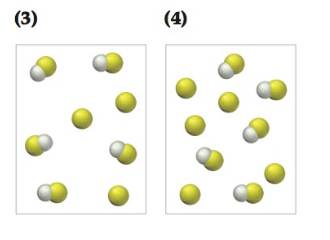
(b) Draw a picture that represents the equilibrium state of solution (1) after the addition of two
(c) Draw a picture that represents the equilibrium state of solution (1) after the addition of two
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Chemistry (7th Edition)
- Acrylic acid is used in the polymer industry in the production of acrylates. Its K, is 5.6 X 10“’. What is the pH of a 0.11 M solution of acrylic acid, CH2CHCOOH?arrow_forwardWrite the equilibrium constant expression, K, for the following reaction. Please enter the compounds in the order given in the reaction. If either the numerator or denominator is blank, please enter 1 CH3 COOH (аq) + H20(1) : H30+(aq) + CH3 COO¯ (aq) K =arrow_forwardWhy does a numerically large K eq mean that the productsare favored in an equilibrium system?arrow_forward
- The ionization constant (Kb) of aniline (C6HSNH2) is 2.35 x 10-5. If 7.285 grams of aniline is dissolved in 100.0 mL of water, what are the equilibrium concentration of all species present?arrow_forwardWrite the equilibrium constant expression, Ke, for the following reaction: If either the numerator or denominator is 1, please enter 1 Pb2*(aq) + Cro,"(aq)PbCrO4(s)arrow_forwardFor the generic equilibrium HA(aq) ⇌ H+ (aq) + A-(aq) ,which of these statements is true?(a) The equilibrium constant for this reaction changes as thepH changes. (b) If you add the soluble salt KA to a solution of HA that is atequilibrium, the concentration of HA would decrease.(c) If you add the soluble salt KA to a solution of HA that is atequilibrium, the concentration of A- would decrease.(d) If you add the soluble salt KA to a solution of HA that is atequilibrium, the pH would increase.arrow_forward
- A buffer is made with sodium acetate (CH3COONa) andacetic acid (CH3COOH); the Ka for acetic acid is 1.80 x 10-5.The pH of the buffer is 3.98. What is the ratio of the equilibriumconcentration of sodium acetate to that of acetic acid?(a) -0.760 (b) 0.174 (c) 0.840 (d) 5.75 (e) Not enough informationis given to answer this question.arrow_forwardWrite the equilibrium constant expression, K, for the following reaction taking place in dilute aqueous solution. HF (aq) + OH- (aq)F- (aq) + H2O (l) K =arrow_forwardMethanoic acid is also called formic acid. It has the chemical formula HCOOH(l). It is a colourless fuming liquid that is mainly used as a preservative. It exhibits the following equilibrium in water:HCOOH(aq) + H2O(l) → HCOO–(aq) + H3O+(aq) 4) The concentration of the hydroxide ion, OH-(aq), in an aqueous solution is 2.5 x 10–3 mol/L. What is the pH of the solution? PLEASE HELP THIS IS VERY URGENTarrow_forward
- Write the equilibrium constant expression, K, for the following reaction. Please enter the compounds in the order given in the reaction. If either the numerator or denominator is blank, please enter 1 K = (CH3)3N(aq) + H₂O(1) — (CH3)3 NH+ (aq) + OH¯(aq)arrow_forwardThe reaction 2CO(g) + O2(g) = 2 CO2(g) is known to be exothermic. According to Le Chatelier's principle, how will this reaction's equilibrium be affected if the reaction temperature is increased? (A) The equilibrium constant will decrease and the reaction will shift to the left. (B) The equilibrium constant will be unchanged, but the reaction will shift to the left. (C) The equilibrium constant will increase and the reaction will shift to the right. (D) The equilibrium constant will be unchanged, but the reaction will shift to the right. (E) The equilibrium concentrations will not be affected.arrow_forwardBe sure to answer all parts. Consider the formation of ammonia in two experiments. (a) To a 1.00-L container at 727°C, 1.30 mol of N₂ and 1.65 mol of H₂ are added. At equilibrium, 0.100 mol of NH3 is present. Calculate the equilibrium concentrations of N₂ and H₂ and find Ke for the reaction: [N₂ leq = [H₂ leq 2 NH3(g) = N₂(g) + 3 H₂(g) Kc = ㅈ = M Kc = (b) In a different 1.00-L container at the same temperature, equilibrium is established with -2 8.34 × 10² mol of NH3, 1.50 mol of N2, and 1.25 mol of H₂ present. Calculate Ke for the reaction: M 3 NH3(g) = N₂(g) + H₂(g) 2 2 C C < Prev *********** 18 of 20 MacBook Pro Search or type URL Next *********** ☆arrow_forward
 Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning


