
Study Guide for Campbell Biology
11th Edition
ISBN: 9780134443775
Author: Lisa A. Urry, Michael L. Cain, Steven A. Wasserman, Peter V. Minorsky, Jane B. Reece, Martha R. Taylor, Michael A. Pollock
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 9, Problem 8IQ
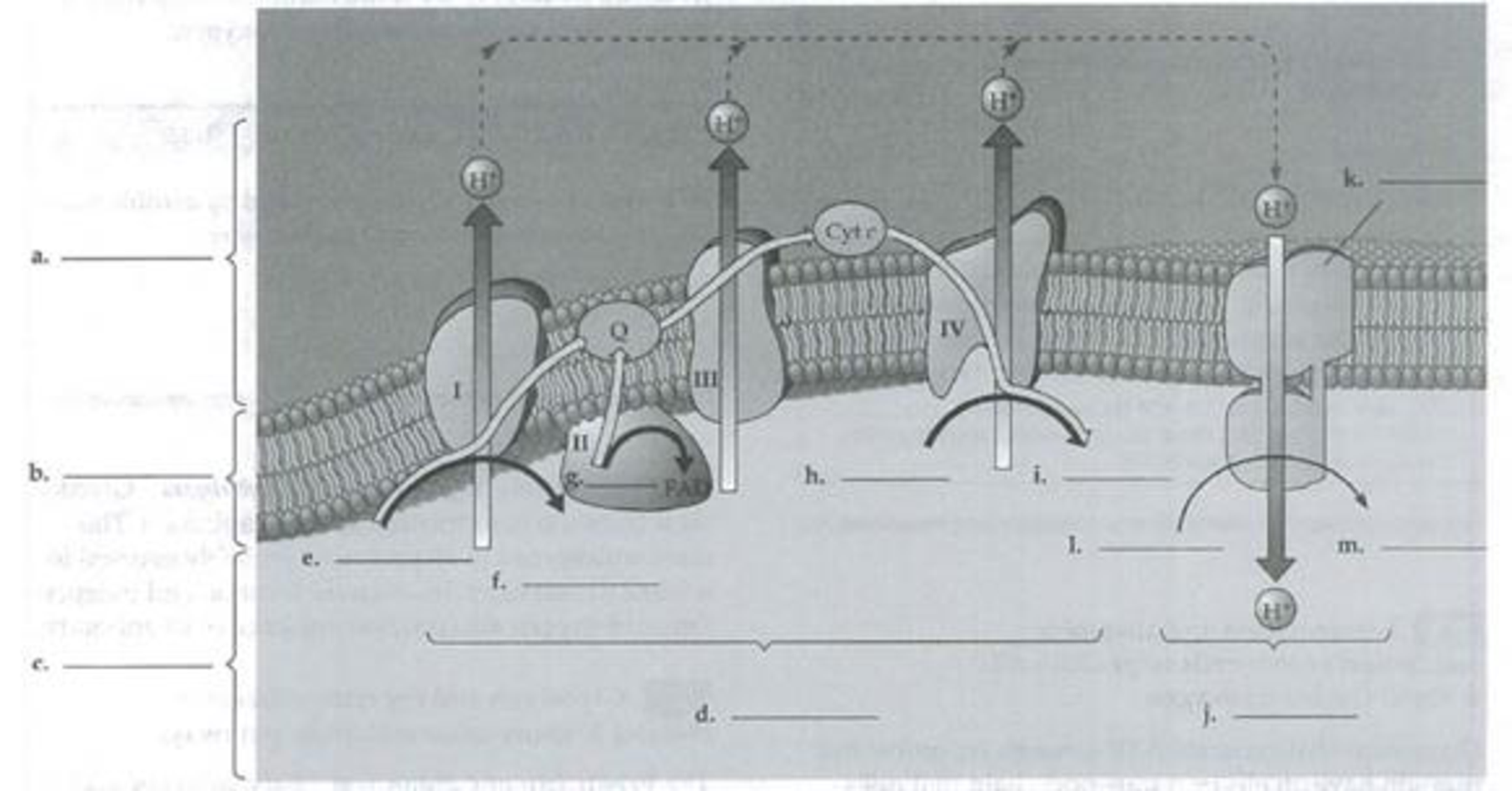
Label the following diagram of oxidative phosphorylation in a mitochondrial membrane.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Fill in the bold using the table.
Because the inner mitochondrial membrane is impermeable to L, when there is an excess of mitochondrial L, it converts to M using a TCA condensation reaction, M crosses to the cytosol using a designated transport system. In the cytosol, M reconverts to L and oxaloacetate. Oxaloacetate is subsequently converted to malate with simultaneous production of N, a prerequisite cofactor for the glycolysis pathway. This reaction is then followed by conversion of N to pyruvate which produces O, a prerequisite cofactor for the fatty acid synthesis pathway.
L
M
N
O
A.
ATP
pyruvate
malate
NADPH
B.
ATP
citrate
ATP
NAD+
C.
acetyl CoA
pyruvate
NAD+
NADH
D.
acetyl CoA
citrate
NAD+
NADPH
E.
glucose
pyruvate
malate
NADP+
Although the outer mitochondrial membrane is permeable to all small molecules, the inner mitochondrial membrane is essentially impermeable in the absence of specific transport proteins. Consider this information answer: If the inner mitochondrial membrane were rendered as permeable as the outer membrane, how would that affect oxidative phosphorylation? Which specific processes would stop and which remain?
Although the outer mitochondrial membrane is permeable to all small molecules, the inner mitochondrial membrane is essentially impermeable in the absence of specific transport proteins. Consider this information answer: The ATP generated by oxidative respiration is used throughout the cell. The majority of ATP production occurs in the mitochondrial matrix. How do you think ATP is made accessible to enzymes in the cytosol and other organelles?
Chapter 9 Solutions
Study Guide for Campbell Biology
Ch. 9 - Fill in the following summary equation for...Ch. 9 - Fill in the appropriate terms in the following...Ch. 9 - a. In the conversion of glucose and O2 to CO2 and...Ch. 9 - a. NAD+ is called a(n) ____________________. b....Ch. 9 - Fill in the three stages of cellular respiration...Ch. 9 - Fill in the blanks in the following summary...Ch. 9 - Fill in the blanks in the following diagram of the...Ch. 9 - Label the following diagram of oxidative...Ch. 9 - Fill in the following tally for the maximum ATP...Ch. 9 - How much more ATP can be generated by aerobic...
Ch. 9 - This chapter describes how catabolic pathways...Ch. 9 - Prob. 2SYKCh. 9 - Fill in the following table to summarize...Ch. 9 - When electrons move closer to a more...Ch. 9 - In the reaction C6H12O6 + 6 O2 6 CO2 + 6 H2O, a....Ch. 9 - In which of the following conversions is the first...Ch. 9 - Some prokaryotes use anaerobic respiration, a...Ch. 9 - Which of the following reactions is incorrectly...Ch. 9 - In which organelle of a plant cell does the citric...Ch. 9 - Which of the following compounds produces the most...Ch. 9 - Aerobic eukaryotes produce CO2 as a by-product...Ch. 9 - Which of the following statements correctly...Ch. 9 - In the electron transport system, H+ ions are...Ch. 9 - When glucose is oxidized to CO2 and water,...Ch. 9 - The energy required for the chemiosmotic synthesis...Ch. 9 - Which of the following statements correctly...Ch. 9 - The citric acid cycle is best described as a. the...Ch. 9 - Fermentation produces less ATP than cellular...Ch. 9 - If muscle cells do not receive enough oxygen from...Ch. 9 - Glucose made from six radioactively labeled carbon...Ch. 9 - Glycolysis is considered one of the first...Ch. 9 - Which of the following substances produces the...Ch. 9 - Fats and proteins can be used as fuel in the cell...Ch. 9 - Which of the following statements is false...Ch. 9 - Brown fat, which is found in newborn infants and...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Similar questions
- In the 1930s, some physicians prescribed low doses of a compound called dinitrophenol (DNP) to help patients lose weight. This unsafe method was abandoned after some patients died. DNP uncouples the chemiosmotic machinery by making the lipid bilayer of the inner mitochondrial membrane leaky to H+ . Explain how this could cause weight loss and death.arrow_forwardAlthough the outer mitochondrial membrane is permeable to all small molecules, the inner mitochondrial membrane is essentially impermeable in the absence of specific transport proteins. Consider this information answer: Present two types of benefits derived from separating the reactions of glycolysis in the cytosol from those that occur during the citric acid cycle in the mitochondrion.arrow_forwardIn the 1930s, some physicians prescribed low doses of a compound called dinitrophenol (DNP) to help patients lose weight. This unsafe method was abandoned after some patients died. DNP uncouples the chemiosmotic machinery by making the lipid bilayer of the inner mitochondrial membrane leaky to H+. Chemical agents that cause this effect are called uncouplers. Explain how this could cause weight loss and also death. Considering the danger, is there any use for compounds like DNP or other uncouplers?arrow_forward
- Describe the molecular mechanisms of mitochondrial ATP synthesis.arrow_forwardThe glycerol-3-phosphate shuttle can transport cytosolic NADH equivalents into the mitochondrial matrix (see Fig. 15.11c). In this shuttle, the protons and electrons are donated to FAD, which is reduced to FADH₂. These protons and electrons are subsequently donated to coenzyme Q in the electron transport chain. End of Chapter Problem 86a How much ATP is generated per mole of glucose when the glycerol-3-phosphate shuttle is used? (Tolerance is +/- 2%) ATP are generated per glucose.arrow_forwardConsider a 24:1 △cis-9 fatty acid in the mitochondrion. For each fatty acid given, determine the following. Total number of ATP deducted Total net ATParrow_forward
- The inner mitochondrial membrane exhibits all of the fundamental characteristics of a typical cell membrane, but it also has several unique characteristics that are closely associated with its role in oxidative phosphorylation. What are these unique characteristics? How does each contribute to the function of the inner membrane?arrow_forwardMatch the following steps of oxidative phosphorylation in increasing order from beginning (1) to end (4) Electrons are transferred to molecules in the beginning of the electron transport chain H+ ions are pumped across the mitochondrial membrane to establish an electrochemical gradient. Electrons are transferred to oxygen, causing it to split and take up H+ ions, which forms water H+ ions flow down the gradient to generate ATParrow_forwardDraw a simple sketch illustrating an inner mitochondrial membrane that is actively involved in chemiosmosis and label the two compartments it separates. Add the ATP synthase complex, indicate the proton gradient, and specify in which compartment ATP is synthesized.arrow_forward
- Label the diagram and explain the salient feature that is happening in each item. For the complexes and the mobile carriers, kindly give their alternative names Electron Transport Chain Intermembrane space Inner mitochondrial membrane Intermembrane space Mitochondrial matrix 1 2 FADH₂ NADH NAD + H Mitochondrial matrix 1. 2. 3. 4. 5. 6. 7. 6 FAD H₂O 2H* + 1/20₂ Inner mitochondrial membrane Name of the Transporter 500 2e- 13040 ALLD LC ATP ADP Essential Featuresarrow_forwardThe maintenance of a proton motive force across the inner mitochondrial membrane is crucial for continued ATP production. Surprisingly, it has been discovered that the inner membranes of certain cells contain proteins, called uncoupling proteins, that are capable of transporting protons from the intermembrane space to the mitochondrial matrix. Why would mitochondria contain transporters that essentially waste energy potential in the proton gradient?arrow_forwardThe fatty acid side chains of the phospholipids in the inner mitochondrial membrane exhibit a large proportion of multiple unsaturations. As a result, will the membrane be hydrophobic or hydrophilic? Also will it be more fluid or more rigid? How do these characteristics help explain the observation that the inner membrane is impermeable to ions such as Mg2+ and Cl–? Hint: what moves these ions through the membrane?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning
Human Physiology: From Cells to Systems (MindTap ...BiologyISBN:9781285866932Author:Lauralee SherwoodPublisher:Cengage Learning

Human Physiology: From Cells to Systems (MindTap ...
Biology
ISBN:9781285866932
Author:Lauralee Sherwood
Publisher:Cengage Learning
Mitochondrial mutations; Author: Useful Genetics;https://www.youtube.com/watch?v=GvgXe-3RJeU;License: CC-BY