
a)
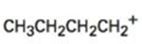
Interpretation:
The structure of more stable carbocation obtainable by the rearrangement of the n-butylcarbocation is to be proposed.
Concept introduction:
The stability of carbocations are in the order tertiary > secondary > primary. A carbocation will rearrange to a more stable carbocation either by a hydride shift or by an alkyl shift from adjacent carbon.
To propose:
The structure of more stable carbocation obtainable by the rearrangement of the carbocation given.
b)
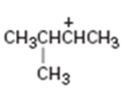
Interpretation:
The structure of more stable carbocation obtainable by the rearrangement of the carbocation given is to be proposed.
Concept introduction:
The stability of carbocations are in the order tertiary > secondary > primary. A carbocation can rearrange to a more stable carbocation either by a hydride shift or by an alkyl shift from adjacent carbon.
To propose:
The structure of more stable carbocation obtainable by the rearrangement of the carbocation given.
c)
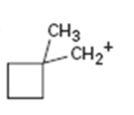
Interpretation:
The structure of more stable carbocation obtainable by the rearrangement of the carbocation given is to be proposed.
Concept introduction:
The stability of carbocations are in the order tertiary > secondary > primary. A carbocation will rearrange to a more stable carbocation either by a hydride shift or by an alkyl shift from adjacent carbon.
To propose:
The structure of more stable carbocation obtainable by the rearrangement of the carbocation given.
Trending nowThis is a popular solution!

Chapter 7 Solutions
Organic Chemistry
- Give the substitution and elimination products for the following reactions, showing the configuration of each product. It develops all the steps of the reaction mechanisms.arrow_forward3. Each of the following carbocations can rearrange to a more stable ion. Propose structures for the likely rearrangement product. a) CH3CHCHCH3 CH3arrow_forwardpredict the product of each of the following reactions. Mono-halogenated products only for any radical halogenation reactions.arrow_forward
- piperidine undergoes the series of reactions shown here, 1,4-pentadiene is obtained as the product. When the four different methyl-substitutedpiperidines undergo the same series of reactions, each forms a different diene: 1,5-hexadiene; 1,4-pentadiene; 2-methyl-1,4-pentadiene; and3-methyl-1,4-pentadiene. Which methyl-substituted piperidine forms which diene?arrow_forwardWhich of the following carbocations would be to rearrange and WHY?arrow_forwardWhich of the following carbocations would you expect to rearrange?arrow_forward
- Two of the carbocations are prone to rearrangement. Show how they might rearrange to more stable carbocationsarrow_forwardWhich of the given reaction schemes would produce the molecule shown below?arrow_forwardWhen piperidine undergoes the series of reactions shown here, 1,4-pentadiene is obtained as the product. When the four different methyl- substituted piperidines undergo the same series of reactions, each forms a different diene: 1,5-hexadiene; 1,4-pentadiene; 2-methyl-1,4-pentadiene; and 3-methyl-1,4-pentadiene. Which methyl-substituted piperidine forms which diene?arrow_forward
- Give the product and mechanism for the following intramolecular reaction. Be sure to include all mechanism arrows, lone pairs, and formal charges. The product is an ether.arrow_forwardIn which of the following carbocations can the rearrangement phenomenon take place and in which cannot? + + + + + ++ Harrow_forwardWrite a step by step mechanism. Includes resonance structures that exist in the transition states.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
