
a)
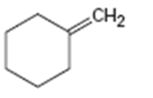
Interpretation:
The major product obtainable from addition of HBr to methylidenecyclohexane is to be predicted.
Concept introduction:
The first step in the addition of HBr with
To predict:
The major product obtainable from addition of HBr to methylidenecyclohexane.
b)
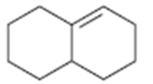
Interpretation:
The major product obtainable from addition of HBr to the octahydronaphthalene is to be predicted.
Concept introduction:
The first step in the addition of HBr with alkenes is the formation of a stable carbocation by the attack of the π electrons of the double bond on the positively polarized hydrogen of HBr eliminating a bromide ion. In the second step the carbocation reacts with the bromide ion to yield the product.
To predict:
The major product obtainable from addition of HBr to octahydronaphthalene.
c)
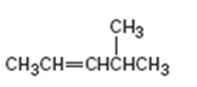
Interpretation:
The major product obtainable from addition of HBr to 4-methyl-2-pentene is to be predicted.
Concept introduction:
In electrophilic addition reactions, the first step is the attack of the π electrons of the double bond on the hydrogen of another reactant to yield a carbocation. One of the carbon in C=C gets attached to hydrogen while the other acquires a positive charge. In the second step, the carbocation formed can rearrange to give another more stable carbocation either by a hydride shift (shift of hydrogen atom with its electron pair) or by an alkyl shift (shift of an alkyl group with its electron pair) between neighboring carbons. In the last step the carbocation produced reacts with a negatively charged ion or eliminates a proton to give the product.
To give:
The major product obtainable from addition of HBr to 4-methyl-2-pentene.
Trending nowThis is a popular solution!

Chapter 7 Solutions
Organic Chemistry
- Identify two alkenes that react with HBr to form 1-bromo-1-methylcyclohexane without undergoing a carbocation rearrangement.arrow_forwardFor the alkene below, write the structures of the products of its reaction and mechanism for each of the following: 1) Reaction with H2 and a Pd catalyst 2) Reaction with Br2 3) Reaction with HBrarrow_forwardAssign Z or E to the following alkenesarrow_forward
- The electrophilic addition of HBr to 3-cyclohexylbut-1-ene gives a mixture of two constitutional isomers. These two isomers can also be produced from 3-cylcohexybut-2-ene, but one of which requires different reaction conditions; the electrophilic addition of HBr to 3-cylcohexybut-2-ene produces one of these isomers while the electrophilic addition of HBr to 3-cylcohexybut-2-ene, in the presence of peroxides, produces the other one. Draw the structure for the isomer product that can result from the reactants 3-cyclohexylbut-1-ene and 3-cylcohexybut-2-ene using different reaction conditions. Part 1 of 2 Click and drag to start drawing a structure. C 8 DY 000 8: >arrow_forwardGive the systematic name for the major product of this reaction (do not specify stereochemistry): CH3 Br2 Ethanolarrow_forwardPredict the products of the following reactions. Include stereochemistry when necessary. For reactions with more than one step show the product formed after each step.arrow_forward
- Which functional group(s) would be added to 1-methylcyclohexene using the reagents below: H3O+ hydrogen hydroxyl ketone and aldehyde bromine and hydroxyl bromine and hydrogen bromine hydrogen and hydroxyl aldehyde ketone Save for Laterarrow_forwardAcid-catalyzed a-halogenation is a useful reaction for synthesis, especially regarding substitution and elimination reactions. Provide the missing structures (A-C) for the following mini road map. H Br₂ H3O+ A E2 | NaNH2 C NaO SN2 Barrow_forwardWhat products would you expect from reaction of the following alkenes with NBS? If more than one product is formed, show the structures of all.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





