
Chemistry In Context
9th Edition
ISBN: 9781259638145
Author: Fahlman, Bradley D., Purvis-roberts, Kathleen, Kirk, John S., Bentley, Anne K., Daubenmire, Patrick L., ELLIS, Jamie P., Mury, Michael T., American Chemical Society
Publisher: Mcgraw-hill Education,
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 5, Problem 25Q
These three compounds all have the same chemical formula of C8H18. The hydrogen atoms and C─H bonds have been omitted for simplicity.
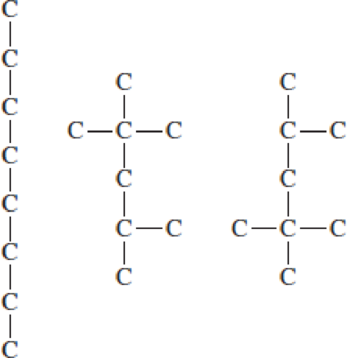
- a. For each compound, draw structural formulas that show the missing H atoms. All should have 18 H atoms.
- b. Which (if any) of these structural formulas are identical?
- c. Draw the structural formulas for any two additional isomers of C8H18.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
a. Give all the structural isomers of the hydrocarbons C5H12.(Hint: 3 isomers)
b. Give all the structural isomers of the compound C3H5Cl which contains one C=C bond. (Hint: 3 isomers)
c. One of the structural isomers in part b shows cis-trans isomerism. Draw and name this pair of isomers.
a. Candle wax contains an alkane with 28 carbon atoms per molecule. What is the molecular formula of this alkane?A. C28H54B. C28H56C. C28H58D. C28H60
b. Which of the following might be the respective relative molecular masses of four consecutive members of a homologous series?(Relative atomic masses: H = 1.0, C = 12.0, O = 16.0)A. 32, 46, 60, 74B. 26, 38, 50, 62C. 28, 38, 48, 58D. 44, 57, 70, 83
1. There are five structural isomers for hexane, C6H₁4. Draw the five structures using simplified
structural formulae. For example, one of the isomers of butane, C4H₁0, could be drawn as
CH3CH₂CH₂CH3. Use the same sort of format for the other questions on this page.
2.
STRUCTURAL ISOMERISM
3.
Draw as many structural isomers as possible for C3H8O.
Draw as many structural isomers as you can for CH₂0 containing a benzene ring.
4. Draw as many structural isomers as you can for C4H8O₂ containing the group
Chapter 5 Solutions
Chemistry In Context
Ch. 5.1 - Prob. 5.1YTCh. 5.1 - Prob. 5.2YTCh. 5.1 - Consult the interactive trends found in the...Ch. 5.1 - Prob. 5.4YTCh. 5.2 - For each of the fuels below, write the balanced...Ch. 5.2 - Prob. 5.6YTCh. 5.3 - Prob. 5.7YTCh. 5.4 - Prob. 5.8YTCh. 5.4 - Prob. 5.9YTCh. 5.4 - Scientific Practices Coal Versus Ethanol On the...
Ch. 5.5 - Prob. 5.12YTCh. 5.5 - Prob. 5.13YTCh. 5.6 - Although power plants require several steps to...Ch. 5.7 - Prob. 5.15YTCh. 5.7 - Prob. 5.16YTCh. 5.7 - An input of energy can be used to decrease entropy...Ch. 5.8 - Prob. 5.18YTCh. 5.8 - Prob. 5.19YTCh. 5.8 - Prob. 5.20YTCh. 5.8 - Prob. 5.21YTCh. 5.10 - Prob. 5.22YTCh. 5.11 - The combustion of one gram of natural gas releases...Ch. 5.11 - a. During the extraction of natural gas, the...Ch. 5.12 - Prob. 5.25YTCh. 5.13 - Prob. 5.26YTCh. 5.13 - Beginning in the 1920s, the octane-booster...Ch. 5.15 - Prob. 5.28YTCh. 5.15 - Prob. 5.29YTCh. 5.16 - Prob. 5.30YTCh. 5.17 - Have you ever been served cherries Jubilee or...Ch. 5.17 - Prob. 5.34YTCh. 5 - Prob. 1QCh. 5 - Prob. 2QCh. 5 - Prob. 3QCh. 5 - Energy exists in different forms in our natural...Ch. 5 - A coal-burning power plant generates electrical...Ch. 5 - Prob. 6QCh. 5 - Prob. 7QCh. 5 - Prob. 8QCh. 5 - Mercury (Hg) is present in trace amounts in coal,...Ch. 5 - Prob. 10QCh. 5 - Here are the condensed structural formulas for two...Ch. 5 - Prob. 12QCh. 5 - Prob. 13QCh. 5 - Consider these three hydrocarbons: At room...Ch. 5 - During petroleum distillation, kerosene and...Ch. 5 - Prob. 16QCh. 5 - a. Write the balanced chemical equation for the...Ch. 5 - Prob. 18QCh. 5 - Prob. 19QCh. 5 - State whether these processes are endothermic or...Ch. 5 - Use the bond energies in Table 5.1 to calculate...Ch. 5 - Use the bond energies in Table 5.1 to calculate...Ch. 5 - Ethanol can be produced by fermentation. Another...Ch. 5 - Here are structural formulas for ethane, ethene...Ch. 5 - These three compounds all have the same chemical...Ch. 5 - Catalysts speed up cracking reactions in oil...Ch. 5 - Explain why cracking is a necessary part of the...Ch. 5 - Consider this equation representing the process of...Ch. 5 - Prob. 29QCh. 5 - Consider these three alcohols: methanol, ethanol,...Ch. 5 - Prob. 31QCh. 5 - Prob. 32QCh. 5 - Prob. 33QCh. 5 - Compare and contrast a molecule of biodiesel with...Ch. 5 - Use Figure 5.6 to compare the energy released for...Ch. 5 - Prob. 36QCh. 5 - The sustainability of burning coal (and other...Ch. 5 - In this chapter, we approximated the chemical...Ch. 5 - Prob. 39QCh. 5 - Compare the processes of combustion and...Ch. 5 - How might you explain the difference between...Ch. 5 - Write a response to this statement: Because of the...Ch. 5 - The concept of entropy and probability is used in...Ch. 5 - Bond energies such as those in Table 5.1 are...Ch. 5 - Use the bond energies in Table 5.1 to explain why...Ch. 5 - Prob. 46QCh. 5 - Prob. 47QCh. 5 - Prob. 48QCh. 5 - Prob. 49QCh. 5 - Prob. 50QCh. 5 - Prob. 51QCh. 5 - Prob. 52QCh. 5 - Prob. 53QCh. 5 - Use a diagram to show the relationship among these...Ch. 5 - On a timescale of a few years, the combustion of...Ch. 5 - Emissions of some pollutants are lower when...Ch. 5 - Although coal contains only trace amounts of...Ch. 5 - Prob. 58QCh. 5 - An article in Scientific American pointed out that...Ch. 5 - C. P. Snow, a noted scientist and author, wrote an...Ch. 5 - Chemical explosions are very exothermic reactions....Ch. 5 - Prob. 63QCh. 5 - Tetraethyllead (TEL) was first approved for use in...Ch. 5 - Tetraethyllead (TEL) has an octane rating of 270....Ch. 5 - Another type of catalyst used in the combustion of...Ch. 5 - Figure 5.8 shows energy differences for the...Ch. 5 - Prob. 68Q
Additional Science Textbook Solutions
Find more solutions based on key concepts
2. Why shouldn’t you work in a laboratory by yourself?
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
Q2. Which statement best defines chemistry?
a. The science that studies solvents, drugs, and insecticides
b. Th...
Introductory Chemistry (5th Edition) (Standalone Book)
Practice Problem 1.22 Which of the following alkenes can exist as cis-trans isomers? Write their structures. Bu...
Organic Chemistry
The structural formula of 1, 2-dimethylbenzene needs to be drawn. Concept introduction: The ring structures of ...
Chemistry: Matter and Change
Draw a Lewis structure for each covalent molecule. a. HBr b. CH3F c. H2O2 d. N2H4 e. C2H6 f. CH2Cl2
Principles of General, Organic, Biological Chemistry
Fully developed conditions are known to exist for water flowing through a 25-nim-diameer tube at 0.01 kg/s and ...
Fundamentals of Heat and Mass Transfer
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 4.61 What is actually measured by the octane ratings of different grades of gasoline?arrow_forward4. Decide whether each statement is true (T) or false (F). Place your answer in the blank space given. a. A hydrocarbon contains only atoms of carbon and hydrogen. b. Alkanes have the general formula C₂H₂- c. In a condensed structural diagram, the symbol "H" is used to show the position of a hydrogen atom. d. A continuous-chain alkane has one or more carbon branches attached to the parent chain. e. In a Lewis dot diagram of a hydrocarbon, each carbon-carbon and carbon-hydrogen bond is represented by a line segment. f. Any branch within branched alkanes is called an alkyl group. 1arrow_forwardWhich element is present in all organic compounds?A. Helium C. CarbonB. Nitrogen D. Oxygen 2. What products are obtained with CH 4 (g) burns completely in an excess ofoxygen? A. CO 2 C. Hydrogen gasB. CO 2 and H 2 O D. Explosion 3. A hydrocarbon molecule is saturated if the molecule contains ___________A. Single covalent bonds, only C. A double covalent bond, onlyB. A triple covalent bond D. Single and double covalent bonds4. A functional group with a carbonyl group functionality is _________________. A. Alcohol C. EsterB. Amine D. Ketone5. Choose the incorrect option regarding Isomerism: A. They differ in both physical and chemical propertiesB. They have the different molecular formulaC. Chain isomers differ in the arrangement of their skeletonD. They have the same molecular formula 6. Which among the following is formed when an alcohol is dehydrated? A. Aldehyde C. AlkeneB. Ketone D. Amine 7. The reaction of water with alkene to produce an alcohol is a/an ______reaction. A. Addition…arrow_forward
- Answer true or false. a. All organic compounds contain one or more atoms of carbon. The majority of organic compounds are built from carbon, hydrogen, oxygen, and nitrogen. By number of atoms, carbon is the most abundant element in the Earth;s crust. Most organic compounds are soluble in water.arrow_forwarda.Give all the structural isomers of the compound C3H5Cl which contains one C=C bond. (Hint: 3 isomers) b.One of the structural isomers in above question shows cis-trans isomerism. Draw and name this pair of isomers.arrow_forward3. Explosives used in mining contain TNT or 2,4,6-trinitrotoluene. a. If the functional group nitro is -NO2, what is the structural formula of TNT? b. TNT is actually a mixture of isomers of trinitrotoluene. Draw two possible isomers.arrow_forward
- 3. An unknown compound 'A' with the formula C4H6 reacts with excess Br2 at room temperature to give 'B' with the formula C4H6Br4. 'A' also react with O3 and H₂O to give C with the formula C4H6O2. Draw the structural formulas for A, B, and C.arrow_forwardIdentify the functional groups present in the structure below. ỌCH3 H3C. Functional Groups a. alkene b. alkyne c. arene d. halide e. alcohol f. ester g. ether h. nitrile i. carboxylic acid j. thiol CH3 N₁ `N Esomeprazole (Nexium) LOCH3arrow_forwardC5H12 has 3 structural isomers C6H14 has 5 structural isomers C7H16 has 9 structural isomers. I get what a structural isomer is, but when I see a statement like the 3 statements above, I wonder if there is a way I can figure out how many structural isomers something has? I'm in CHEM 2. We didnt talk about that but when I see a slide he has telling me this, and I am wondering what to take from the slide...hmmm.arrow_forward
- II. Cite the importance of organic chemistry in various fields like industry, health, and medicine.arrow_forwardConsider methane, CH₄, and hydrogen, H₂, as possible fuel sources a. Describe how methane and hydrogen could be obtained. Which of these methods do you think is less expensive? Explain. b. Which fuel do you think is more environmentally friendly? Explain.arrow_forward8. In a reduction reaction a. a carbon will form fewer bonds to carbon. b. a carbon will form fewer bonds with oxygen or more bonds to hydrogen. c a carbon will form more bonds with oxygen or fewer to hydrogen. d. two small molecules combine to form a large molecule. e. two large molecules react to form a large molecule and a small molecule. 9. What is an addition reaction? a. One in which atoms or groups of atoms are removed from an organic molecule and a double bond is formed. b. One in which an atom or small group replaces another atom or small group on the organic molecule. c. One in which atoms from a small molecule react with a double or triple bond in an organic molecule to become part of the molecule. d. One in which two large molecules combine to form one larger molecule and one small molecule, usually water. 10. What results when a secondary alcohol is oxidized? a. A ketone b. An amine c. An aldehyde d. An acid e. No reaction 11. Which type of reaction will an alkene not…arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:9781337398909
Author:Lawrence S. Brown, Tom Holme
Publisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Chapter 4 Alkanes and Cycloalkanes Lesson 2; Author: Linda Hanson;https://www.youtube.com/watch?v=AL_CM_Btef4;License: Standard YouTube License, CC-BY
Chapter 4 Alkanes and Cycloalkanes Lesson 1; Author: Linda Hanson;https://www.youtube.com/watch?v=PPIa6EHJMJw;License: Standard Youtube License