
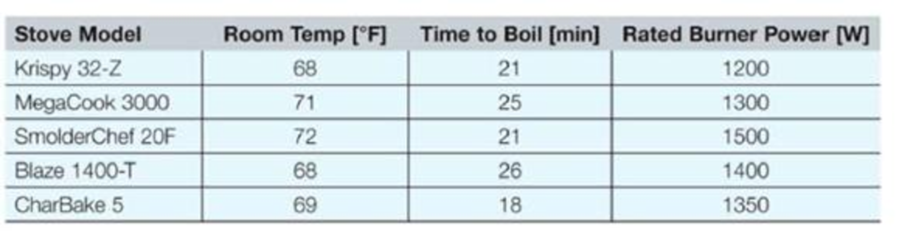
You are part of an engineering firm on contract with the U.S. Department of Energy’s Energy Efficiency and Renewable Energy task force to develop a program to help consumers measure the efficiency of their home appliances. Your job is to write a program that measures the efficiency of stove-top burners. Before using your program, the consumer will place a pan of room-temperature water on the stove (with + gallon of water), record the initial room temperature in units of degrees Fahrenheit, turn on the burner, and wait for it to boil. When the water begins to boil, the consumer will record the time in units of minutes it takes for the water to boil. Finally, the consumer will look up the power for the burner provided by the manufacturer.
The output of your program should look like the following output, where the highlighted values are example responses typed by the user into your program. Note that your code should line up the energy and power calculations as shown. In addition, your code must display the efficiency as a percentage with one decimal place and must include a percent symbol.
Sample Input/Output
Household Appliance Efficiency Calculator: Stove
Type the initial room temperature of the water [deg E]: 68 Type the time it takes the water to boil [min]: 21 Type the brand name and model of your stove: Krispy 32-Z Type the power of the stove-top burner [W]: 1200
Energy required: 1267909 J
Power used by burner: 1006 W
Burner efficiency for a Krispy 32-Z stove: 83.9
Want to see the full answer?
Check out a sample textbook solution
Chapter 18 Solutions
Thinking Like an Engineer: An Active Learning Approach (3rd Edition)
- QUESTION 10 A Watt (W) is defined as one Joule (J) of energy per second (s): 1 W = 1 J/s. It is also possible to calculate the watts of an electric device by multiplying the voltage supplied, in Volts (V), by the current drawn, in Amps (A): 1 W = 1 V x 1 A. Consider an electric car with a 75 kWh (kilowatt- hour) battery. If the car consumes on average 230 Wh/mile, what is the maximum range of the car in miles? Enter your answer to one decimal place using rounding.arrow_forwardPosted below is a picture of a data table and a diagram of an experiment. In the experiment you rolled a marble down a ruler ramp off the end of a coffee table to observe projectiles and conservation of energy. You do 3 trials for each initial height. Each height is measured in meters. h= initial height of marble off of table Y= height of table off floor X= horizontal distance marble goes from edge of table to where it strikes the floor a) use the table to find information you need to solve for theoretical range and % of error in measured range.arrow_forward1-heat is defined as the average kinetic energy of the atoms and molecules that make up a substance and temperature is the amount of thermal energy transported2- heat is defined as the total kinetic energy of the atoms and molecules that make up a substance and temperature is the transport of pressure energy due to the temperature gradient3- Heat is defined as the average kinetic energy of the atoms and molecules that make up a substance and temperature is a measure of the equivalent kinetic energy of the individual molecules or atoms4- Heat is defined as the total kinetic energy of the atoms and molecules that make up a substance and temperature is a measure of the total kinetic energy of the individual molecules or atoms5- Heat is defined as the total kinetic energy of the atoms and molecules that make up a substance and temperature is a measure of the average kinetic energy of the individual molecules or atoms choose only one of the above alternatives as correctarrow_forward
- Solve question 2 based on the answer of question 1. You have a natural gas furnace in your home that used 78,500 cubic feet of natural gas for heating last winter. Your neighbor has a furnace that burns heating oil and used 516 gallons of heating oil last winter. You can convert the natural gas and heating oil consumption data into Btu to determine which home used more energy for heating. Natural gas BTU: 1,028 Btu per cubic foot Oil BTU: 138,590 Btu per gallon Natural Gas BTU= 80,698,000 Oil BTU = 71,512,440 The home that used a natural gas furnace used more energy for heating. 2.- You need a new furnace for your home, and you are comparing systems that use natural gas and heating oil. One factor to consider is the cost of the fuel. You can compare the price of the fuels on an equal basis by dividing the price per unit of the fuels by the Btu content per unit of the fuels to get a price per million Btu. Assume Natural gas price = $10.50 per thousand cubic…arrow_forward1. Energy consumption in a country from 2001 to 2011 increased from 1041.3 million to 1526.1 million BOE (barrels of oil equivalent). If the high calorific value (HHV) of the oil is 141,000 kJ/gallon what is the increase in energy consumption over 10 years in Joule units. What is the rate of energy consumption per year? 2. Based on data on the rate of energy consumption per year from question no. 1, if the growth rate energy 2.5% per year, calculate the doubling time (td) and what is the approximate rate of energy growth in 2021.arrow_forwardHi, I need help with the first part of the problem below because I'm very confused about how P1 and P2 should be calculated. If you look at my notes to solve the problem there is already a formula in place as I always thought the Patm should also be multiplied by the Area in the numerator, but it's not if I look at this specific tutorial solution given by my course but it's not explained why. I have done a while ago a very similar problem with using that formula in my notes and it gave me the right results, but it's not working for this one. Could you please help me understand why as I have a test coming soon? Figure Q3 (see image attached) shows a cylinder and pistonenclosing air, the movement of the pistonbeing restrained by a compression spring ofstiffness 20 kN/m. The air is heated andexpands, the piston moving 0.3 m. Thefree length of the spring is 1.0 m.Calculate the work done by the air duringthe process.If the pressure , volume and internal energyof air are related by the…arrow_forward
- show schematic drawings, conversions, units and box in your final answers Determine the amount of energy (kJ) that needs to be transferred to the water to raise its temperature to 140°F if a water heater is initially filled with 50 gallons of water at 64.4°F.arrow_forwardRow indexing: Snow fall records stateSnowRecord contains the amount of snow fall (in cm) for various cities. Rows represent a city and columns represent the snow fall for a given month. Assign citySnow Record with all elements of row rowNum. Ex: If stateSnowRecord is [17.3, 20.3; 4.8, 6.2] and rowNum is 2, then citySnowRecord is [4.8, 6.2]. Function > 6 % 7 % 8 9 10 11 12 13 end function citySnowRecord = Get CitySnowFall (stateSnowRecord, rowNum) 2% Get CitySnow Fall: Returns all snow fall values for city in row rowNum % 4 % Inputs: stateSnowRecord matrix of snow fall records for cities for various months rowNum specified row of stateSnowRecord to return stateSnowRecord = [17.3, 20.3; 4.8, 6.2] Outputs: citySnowRecord - all snow fall values for a given city % Assign citySnowRecord with all elements of a row rowNum. citySnowRecord = stateSnowRecord (rowNum, :); Code to call your function > 1 GetCitySnow Fall([17.3, 20.3; 4.8, 6.2], 2) Previous Assessment: 1 of 2 Tests Passed (50%) Save…arrow_forwardBox the final answer. Write legibly. An adiabatic room (with dimensions, LxWxH) with the smallest side equal to 5.25 meters initially contains air at 18.25ºC and 1.025 bar. A total of 52.5 KW of heat have been added to double its temperature. Calculate the total time elapsed in minutes to reach that final temperature if the dimensions of the room have a ratio of 3:2:1, respectively.arrow_forward
- Read the question carefully and match the answer with its respective question: It has been reported that, before 10years, the Heat Rate of natural gas was 8185. It has been observed that, due to the use of modern technology, in last 10years, the efficiency of natural gas-based power plants has been increased by 20%. As per the report, what is the new %age efficiency? Choose. With reference to the new efficiency what would be the new recorded value of the heat rate of natural gas? Choose.arrow_forward) Prof. K. took a Tesla Model S for a test drive. The car weighs 4,500 lb and can accelerate from | O to 60 mph in 2.27 seconds. The car has a 581 kW battery pack. Help Prof. K. determine the efficiency of the vehicle [Ans. to Check 55.5%). Note: The equation we learned in class P= F v ONLY APPLIES if velocity is constant. Use the steps below to guide your thought process. Develop a symbolic relationship for the force required to move the car forward. Compute the work done by the car (in Ib-ft). Calculate the power required to do this work (in kW). Ey el mp cul 2) After playing a game of pinball, you decide to do some calculations on the launching 9:09 AM P Type here to search 33°F Mostly sunny 3/7/2022 PrtScn Tnd Homearrow_forwardA Team of Engineers asked for an internal combustion engine to use it in a designed car. Your role is to describe the operation sequence of different types of available engines, explain their mechanical efficiency, and deliver a detailed technical report to show your approach in solving and discussing the following tasks and issues. You must follow the following steps to help the team: STEP 1 Describe the operational sequence of four-stroke spark ignition and four-stroke compression ignition engines with the aid of sketches by constructing simple sketch representing the operation and plotting the P-V diagrams for each process during the cycle to show the following: The input and output heat and net output work The air-fuel mixture intake and exhaust gasses The spark plug when it is in the active modearrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





