
Review the oxidation reactions using
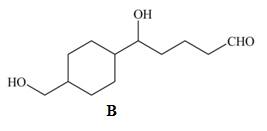
a.
b.
c. PCC
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Organic Chemistry (6th Edition)
- 19. Choose the correct reagents/reactants to synthesize the following compound. 1. NaH 1. NaH A. OH 2. OH 2. Br Br 3. H3O* С. 1. NaH 1. NaH HO 2. HO 2. Br Br 3. H;0* B. D.arrow_forwardWhat is the product of the following reaction? Ion OH 1. Br2, HOẶC 2. pyridine Br = = Br IV Brarrow_forwardDescribe the preparation and storage of each reagents. d. 2 L of 0.1 M NaOH e. 1.5 L of 0.04 M KMnO4 f. 3 L of 1 M H2SO4 g. 500 mL of 0.1 M HCl Conclusion.arrow_forward
- Why is 1.5 M of H2SO4 more dangerous and hazardous than 2.5 M CH3COOHarrow_forwardGive the organic product formed. x.8—- X + OA. I B. II heat x x x x ? = IV 0 40 H US 0arrow_forward7. Use the curved arrow notations to show the flow of electrons in the given reactions. 4. a. b. OH OH 4. → OH. ofarrow_forward
- MgBr 1. PhCHO, then sat. NH4Cl(aq) 2. Jones reagent 3. Saegusa-Ito oxidation 4. Me₂CuLi, then workup 5. Ph3P=CHPharrow_forwardThe compound 1-chloropropane immediately formed a white precipitate when NaI in acetone was added. What is the identity of the white precipitate? a. 1-chloropropane b. Sodiumchloropropane c. NaCl d. NaIarrow_forwardwhich compound listed below will dissolve best in H2O ? EXPLAIN your choice . A. CH2O B. CO2 C. O2arrow_forward
- 8. Fehling's reagent is:A) A solution of copper sulfate in sulfuric acidB) A mixture of a solution of copper sulfate and tartaric acid with the addition of sodium hydroxideC) A mixture of a solution of copper sulfate in sulfuric acid and potassium hydrogen tartrate with sodium hydroxideD) A solution of tartaric acid in potassium hydroxidearrow_forward1. Dimethyl disulfide, CH,S-SCH, found in the vaginal secretions of female hamsters, acts as a sexual attractant for the male hamster. Write an equation for its synthesis from methanethiol. 2. Write an equation for the reaction of ethylene oxide with a. I mole of HCI b. excess HCI c. phenol +H d. phenylmagnesium bromidearrow_forward-Which substance has the greatest tendency to participate in a chemical reaction with small chain alcohol in an acidified chemical environment? a. Substance A b. Substance B c. Substance C d. Substance D e. Substance E -Which substance is expected to turn blue litmus paper into red color? a. Substance A b. Substance B c. Substance C d. Substance D e. Substance E -The physical properties of the substances are influenced by intermolecular forces of attraction. Which substance(s) are predominantly influenced by the intermolecular association of the molecules via hydrogen bonding? a.Substance A only b. Substance A and C c. Substance A, B, and C d.Substance A, B, C, and D -Aside from London dispersion forces, which substance do you expect dipole interactions to be effective? a. Substance A and B b. Substance B and D c. Substance A and C d. Substance B and E -Which substance will exhibit the lowest melting point? a. Substance A b. Substance B c. Substance C d. Substance D e. Substance E…arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





