
Organic Chemistry (6th Edition)
6th Edition
ISBN: 9781260119107
Author: Janice Gorzynski Smith
Publisher: McGraw Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 17.2, Problem 2P
Which carbonyl groups in the anticancer drug taxol (Section
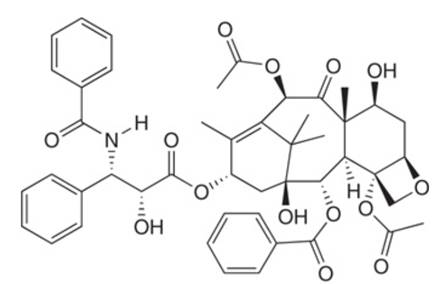
taxol
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Ibufenac, a para-disubstituted arene with the structure HO2CCH2C6H4CH2CH(CH3)2, is a much more potent analgesic than aspirin, but it was never sold commercially because it caused liver toxicity in some clinical trials. Devise a synthesis of ibufenac from benzene and organic halides having fewer than five carbons.
Which carbonyl groups in the anticancer drug taxol (Section 5.5) will undergo nucleophilic addition and which will undergo nucleophilic substitution?
Muscalure, the sex pheromone of the common housey, can be prepared by a reaction sequence that uses two nucleophilic substitutions. Identify compounds A–D in the following synthesis of muscalure.
Chapter 17 Solutions
Organic Chemistry (6th Edition)
Ch. 17.1 - Prob. 1PCh. 17.2 - Which carbonyl groups in the anticancer drug taxol...Ch. 17.2 - Prob. 3PCh. 17.4 - Problem 20.4 What alcohol is formed when each...Ch. 17.4 - Problem 20.5 What aldehyde or ketone is needed to...Ch. 17.5 - Problem 20.8 Draw the products formed (including...Ch. 17.7 - Problem 20.10 Draw a stepwise mechanism for the...Ch. 17.7 - Prob. 10PCh. 17.7 - Problem 20.12 Draw the products formed from ...Ch. 17.7 - Prob. 12P
Ch. 17.7 - Prob. 13PCh. 17.8 - Prob. 14PCh. 17.8 - Problem-20.16 Review the oxidation reactions using...Ch. 17.9 - Problem-20.17 Write the step(s) needed to convert ...Ch. 17.9 - Prob. 18PCh. 17.10 - Problem 20.21 Draw the product of each reaction.
...Ch. 17.10 - Problem 20.22 Draw the products (including...Ch. 17.11 - Problem 20.23 What Grignard reagent and carbonyl...Ch. 17.11 - Problem 20.24 Linalool (the Chapter 9 opening...Ch. 17.11 - Problem 20.25 What Grignard reagent and carbonyl...Ch. 17.12 - Prob. 24PCh. 17.13 - Problem 20.28 What ester and Grignard reagent are...Ch. 17.13 - Prob. 27PCh. 17.13 - Problem 20.30 What reagent is needed to convert ...Ch. 17.13 - Prob. 29PCh. 17.14 - What carboxylic acid formed from each alkyl halide...Ch. 17 - 20.37 Devise a synthesis of each alcohol from...Ch. 17 - 20.38 Draw the products formed when pentanal is...Ch. 17 - 20.39 Draw the product formed when is treated...Ch. 17 - The stereochemistry of the products of reduction...Ch. 17 - Prob. 40PCh. 17 - 20.42 Draw the products or each reduction...Ch. 17 - 20.44 Draw all stereoisomers formed in each...Ch. 17 - 20.54 Draw a stepwise mechanism for the following...Ch. 17 - Prob. 57PCh. 17 - Prob. 58PCh. 17 - 20.57 What ester and Grignard reagent are needed...Ch. 17 - 20.58 What organolithium reagent and carbonyl...Ch. 17 - 20.59 What epoxide and organometallic reagent are...Ch. 17 - Prob. 62PCh. 17 - 20.69 An unknown compound A (molecular formula )...Ch. 17 - 20.70 Treatment of compound C (molecular formula )...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A key step in a synthesis of the antimalarial drug quinine involves an intramolecular nucleophilic substitution that converts A to B. Draw the structure of B and give the reagents needed to convert B to quinine.arrow_forwardWhat neutral nucleophile is needed to convert dihalide A to ticlopidine, an antiplatelet drug used to reduce the risk of strokes?arrow_forwardFluorination of a benzene ring can be accomplished with Selectfluor, a reagent that contains a fluorine bonded to a positively charged nitrogen atom. Fluorination is a useful reaction because several common drugs, such as the cholesterol-lowering drug atorvastatin, contain a fluorine bonded to an aromatic ring. Assuming that fluorination is analogous to other examples of electrophilic aromatic substitution, draw a stepwise mechanism for the following reaction.arrow_forward
- Acyclovir is an effective antiviral agent used to treat the herpes simplexvirus. (a) Draw the enol form of acyclovir, and explain why it is aromatic.(b) Why is acyclovir typically drawn in its keto form, despite the fact thatits enol is aromatic?arrow_forwardDraw the products formed when p-methylaniline (p-CH3C6H4NH2) istreated with following reagent. Part (b), then CH3COCl, AlCl3arrow_forwardRank the compounds in each group in order of increasing reactivity toward nucleophilic attack.arrow_forward
- Why is it not advisable to use aqueous hydrochloric acid in a Grignard reaction of a ketone? A) The Grignard reagent will react with the acid and cannot react with the ketone. B) The ketone will be protonated and will become unreactive. C) The ketone will form an unreactive enol. D) The Grignard reagent won't dissolve in aqueous solutionsarrow_forwardThe compound eutypine is an antibacterial agent isolated from the fungus Eutypa lata. This fungus results in a disease common to vineyards called eutyposis. Give a sequence of reactions that will take the following reactant and give eutypine when the other reactants used in the sequence are acetylene and acetone.arrow_forwardIdentify the missing reagents a-f in the following scheme:arrow_forward
- Fill in the missing reagents a-e in the following scheme:arrow_forwardMuscalure, the sex pheromone of the common housefly, can be prepared by a reaction sequence that uses two nucleophilic substitutions. Identify compounds A–D in the following synthesis of muscalure.arrow_forwardCoumarin, a naturally occurring compound isolated from lavender, sweet clover, and tonka bean, is made in the laboratory from o-hydroxybenzaldehyde by the reaction depicted below. Draw a stepwise mechanism for this reaction. Coumarin derivatives are useful synthetic anticoagulants.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Characteristic Reactions of Benzene and Phenols; Author: Linda Hanson;https://www.youtube.com/watch?v=tjEqEjDd87E;License: Standard YouTube License, CC-BY
An Overview of Aldehydes and Ketones: Crash Course Organic Chemistry #27; Author: Crash Course;https://www.youtube.com/watch?v=-fBPX-4kFlw;License: Standard Youtube License