
Concept explainers
Interpretation:
The dicarboxylic acid and
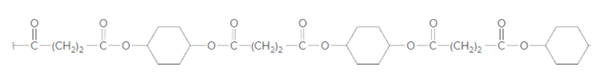
Concept Introduction:
On the basis of type of polymers, polymerization reaction can be two types; addition polymerization and condensation polymerization. The addition polymerization occurs between monomer units with unsaturated bonds between carbon atoms. For example; polymerization of
Condensation polymerization occurs with those monomers which have some
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
General, Organic, and Biological Chemistry - 4th edition
- What are hemiacetals and acetals, and how are they prepared?arrow_forwardHow many esters can be formed by ethylene glycol with formic and acetic acids? (One or both of the –OH groups of ethylene glycol can react.)arrow_forwardState the alcohol and carboxylic acid required to form octyl butanoate:arrow_forward
- Draw the structure if the following compounds. benzoic propanoic anhyydride octyl butanoate 30methylhexanoyl chloridearrow_forwardWhich polymer has a carboxylic acid functional group? A) Nomex B) Polyvinyl acetate C) Rubber D) Polyacrylate E) Polyethylene glycolarrow_forwardWhat is the general name of the organic species that forms when a carboxylic acis is reacted with a strong base?arrow_forward
- How may acids be obtained from alcohols and aldehydes?arrow_forwardWhat polyamide is formed from each monomer or pair of monomers?arrow_forward1) To identify which contains more acid vinegar or lemon juice neutralization with sodium hydroxide can be used. Explain how. 2) The long filaments making up the thread of woven synthetic fabrics are likely polyesters. Describe the special characteristic that the alcohol and carboxylic acid must have so that they can react with each other to produce such long filaments.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





