
Concept explainers
(a)
Interpretation:
Acceptable name for the following compound should be determined:
HCONH2
Concept Introduction:
All three carboxylic acids, esters and amides contain carbonyl group (carbon atom connects to oxygen atom via double bond).
In carboxylic acids, there has a carboxylic group (COOH).
In esters, there has an alkoxy group (OR') bonded to the carbonyl carbon.
In amides, there has a nitrogen atom bonded to the carbonyl carbon.
(b)
Interpretation:
Acceptable name for the following compound should be determined:
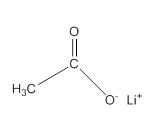
Concept Introduction:
All three carboxylic acids, esters and amides contain carbonyl group (carbon atom connects to oxygen atom via double bond).
In carboxylic acids, there has a carboxylic group (COOH).
In esters, there has an alkoxy group (OR') bonded to the carbonyl carbon.
In amides, there has a nitrogen atom bonded to the carbonyl carbon.
(c)
Interpretation:
Acceptable name for the following compound should be determined:
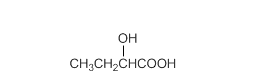
Concept Introduction:
All three carboxylic acids, esters and amides contain carbonyl group (carbon atom connects to oxygen atom via double bond).
In carboxylic acids, there has a carboxylic group (COOH).
In esters, there has an alkoxy group (OR') bonded to the carbonyl carbon.
In amides, there has a nitrogen atom bonded to the carbonyl carbon.
(d)
Interpretation:
Acceptable name for the following compound should be determined:
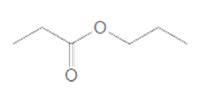
Concept Introduction:
All three carboxylic acids, esters and amides contain carbonyl group (carbon atom connects to oxygen atom via double bond).
In carboxylic acids, there has a carboxylic group (COOH).
In esters, there has an alkoxy group (OR') bonded to the carbonyl carbon.
In amides, there has a nitrogen atom bonded to the carbonyl carbon.
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
General, Organic, and Biological Chemistry - 4th edition
- 1. CH₂CO3H 2. EtOH, H* ?arrow_forwardReacciones Utilizando Cloruros de Acilo 1. LiAlH4 2. HCI 5% H₂O ру OH Et3N OH 2 CH3NH2 CI Et3N 2 NH ру H₂O OH- OH HOarrow_forward16. What is one use for sodium and potassium salts of long-chain carboxylic acids? Oa. food preservative b. athletes foot treatment C. soaps d. pH buffer e. blood anticoagulantarrow_forward
- 15 7. a. 1. CH,CO₂Et, NaOH, EtOH then H₂O* 2. H3PO4, H₂NEt 3. LiAlH, THF ??? OH NH ZH H b. OH IN C. a. NH d. от NHarrow_forwardWhat is the major product to the following reaction? HCI H. H. CI H. KD CI H.arrow_forwardMCQ 169: At temperatures above 1500 °C, hydrogen fluoride (HF) and hydrogen chloride (HCI) are A. decomposed B. not decomposed C. stable D. meltarrow_forward
- H A 1. KCN, H,O 2. NaBH4, MeOH 3. cyclopentanone TSOH, benzene heat o look for B Ph Ph- C D Ph OHarrow_forwardwhich has the greatest acidity? a. 2-hexanol b. valeric acid c. naphtol d. ethyl propionate which produces effervescence when dissolved in 10% sodium bicarbonate? a. 2-hexanol b. valeric acid c. napththol d. ethyl propionatearrow_forwardProvide the starting material for the following reaction: OH A. O NaOH H₂O B. C. D. I I H H Harrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





