
General, Organic, and Biological Chemistry - 4th edition
4th Edition
ISBN: 9781259883989
Author: by Janice Smith
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 16, Problem 84P
Interpretation Introduction
(a)
Interpretation:
The
Concept Introduction:
Functional groups in a molecule are the substituents that are responsible for the characteristic
Interpretation Introduction
(b)
Interpretation:
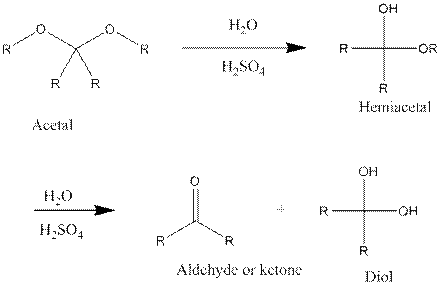
The products which are formed during the hydrolysis of safrole with water and sulfuric acid should be determined.
Concept Introduction:
When an acetalreacts with water in the presence of strong acid that is
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
This carboxylic salts are effective ingredient against yeast and molds in beverages, jams, pie fillings and ketchup
a. benzoates
b. sorbates
c. acetates
d. propionates
1. What is the purpose of Salicylic acid in the synthesis of aspirin?
a. Electrophile
b. Catalyst
c. Nucleophile
d. Side product
2. What is the purpose of Salicylic acid in the synthesis of Oil of Wintergreen?
a. Electrophile
b. Catalyst
c. Nucleophile
d. Side product
Draw the structure corresponding to each name. a. propyl formate b. butyl butanoate c. heptyl benzoate d. N-ethylhexanamide e. N-ethyl- N-methylheptanamide
Chapter 16 Solutions
General, Organic, and Biological Chemistry - 4th edition
Ch. 16.1 - Prob. 16.1PCh. 16.1 - Draw the structure of the three constitutional...Ch. 16.2 - Give the IUPAC name for each aldehyde. a. (...Ch. 16.2 - Give the structure corresponding to each TUPAC...Ch. 16.2 - Prob. 16.4PCh. 16.2 - Prob. 16.2PPCh. 16.2 - Give the structure corresponding to each name. a....Ch. 16.3 - Which compound in each pair has the higher boiling...Ch. 16.3 - Acetone and progesterone are two ketones that...Ch. 16.4 - Prob. 16.8P
Ch. 16.5 - What product is formed when each carbonyl compound...Ch. 16.5 - Prob. 16.4PPCh. 16.5 - Prob. 16.9PCh. 16.6 - Prob. 16.5PPCh. 16.6 - Prob. 16.10PCh. 16.6 - Prob. 16.11PCh. 16.7 - Prob. 16.12PCh. 16.8 - Prob. 16.6PPCh. 16.8 - Prob. 16.7PPCh. 16.8 - Prob. 16.13PCh. 16.8 - Prob. 16.8PPCh. 16.8 - Label the three acetalsin solanine, the toxic...Ch. 16.8 - Prob. 16.14PCh. 16.8 - Prob. 16.10PPCh. 16 - Prob. 15PCh. 16 - Prob. 16PCh. 16 - Prob. 17PCh. 16 - Prob. 18PCh. 16 - Prob. 19PCh. 16 - Prob. 20PCh. 16 - Prob. 21PCh. 16 - Prob. 22PCh. 16 - Prob. 23PCh. 16 - Prob. 24PCh. 16 - Give an acceptable name for each ketone. a. b. c....Ch. 16 - Prob. 26PCh. 16 - Prob. 27PCh. 16 - Draw the structure corresponding to each name. a....Ch. 16 - Prob. 29PCh. 16 - Prob. 30PCh. 16 - Prob. 31PCh. 16 - Prob. 32PCh. 16 - Draw out the structure of benzaldehyde, including...Ch. 16 - Prob. 34PCh. 16 - Which compound in each pair has the higher boiling...Ch. 16 - Prob. 36PCh. 16 - Prob. 37PCh. 16 - Prob. 38PCh. 16 - Prob. 39PCh. 16 - Prob. 40PCh. 16 - Prob. 41PCh. 16 - Prob. 42PCh. 16 - Prob. 43PCh. 16 - Prob. 44PCh. 16 - Prob. 45PCh. 16 - Consider the following ball-and-stick model....Ch. 16 - Prob. 47PCh. 16 - Prob. 48PCh. 16 - Prob. 49PCh. 16 - Prob. 50PCh. 16 - Prob. 51PCh. 16 - Prob. 52PCh. 16 - Prob. 53PCh. 16 - Prob. 54PCh. 16 - Prob. 55PCh. 16 - Prob. 56PCh. 16 - Prob. 57PCh. 16 - Prob. 58PCh. 16 - Prob. 59PCh. 16 - Label the functional group(s) in each compound as...Ch. 16 - Prob. 61PCh. 16 - Prob. 62PCh. 16 - What acetal is formed when each aldehyde or ketone...Ch. 16 - What acetal is formed when each aldehyde or ketone...Ch. 16 - Prob. 65PCh. 16 - Prob. 66PCh. 16 - Prob. 67PCh. 16 - Prob. 68PCh. 16 - Prob. 69PCh. 16 - Prob. 70PCh. 16 - Prob. 71PCh. 16 - Prob. 72PCh. 16 - Prob. 73PCh. 16 - Answer each question about phenylacetaldehyde,...Ch. 16 - Prob. 75PCh. 16 - Prob. 76PCh. 16 - Prob. 77PCh. 16 - Prob. 78PCh. 16 - Three constitutional isomers of molecular formula...Ch. 16 - Identify A—C in the following reaction sequenceCh. 16 - Androsterone is a male sex hormone that controls...Ch. 16 - Prob. 82PCh. 16 - Prob. 83PCh. 16 - Prob. 84PCh. 16 - Prob. 85PCh. 16 - Paraldehyde, a hypnotic and sedative once commonly...Ch. 16 - Prob. 87PCh. 16 - Prob. 88PCh. 16 - Prob. 89CPCh. 16 - Prob. 90CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What kind of solvent ingredients is usually used in the concentrations of 4-10 percent in skin care products and their function is to soften skin cells and to lessen wrinkles? A. Ethly acetate B. Alpha hydroxyl acids C. Phenols and phenol derivatives D. Aliphatic alcoholsarrow_forward1. What are the substances present in the label of formaldehyde (formalin)? 2. What is the percentage composition of acetone in the nail polish remover? 3. According to your interview, what is/are the uses of formalin and acetone? A. Uses of Formalin- B. Uses of Acetone - 4. What type of bond is present in the two structures? 5. Why do you think aldehydes and ketones belong to the group of carbonyl containing compounds?arrow_forward1. Give the product of this reaction A. Propanal B. Propanol C. Propan-2-olD. Acetic acid E. Propanoic acidarrow_forward
- FLAVORANTS: Identify the aldehyde and ketone-containing flavoring compound from each following sources. Choose your best answer from the choices below A. 2-Octanone B. Citral C. Benzaldehyde D. Vanillin E. Cinnamaldehyde F. a-Damascone 1. Вerry 2. Mushroom 3. Lemongrass 4. Cinnamon 5. Almonds Idchyde or ketone formed froarrow_forward1. Differentiate Ethyl alcohol from methyl alcohol. 2. Compare aldehydes and ketones as to (Use acetaldehyde and acetone as examples). A. Reaction with cone. NaOH, heated B. Reaction with Tollen’s reagentarrow_forward15. Circle the structure of limonene which you isolated from orange peels. a.c.d. b.arrow_forward
- Glycerol contains: a. oxygens which are each bonded to two alkyl groups b. oxygens single-bonded to primary and secondary carbons c. Oxygens double-bonded to carbon, with alkyls on both sides d. Oxygens double-bonded to carbon, with alkyls on one side only e. Oxygens double-bonded to carbon, with an alkyl on one side and an --OH on the other sidearrow_forwardWhat are the best starting material and reagents needed to make N-methylpropanamide? a. propanol and H2CrO7 b. propanoic acid, methylamine c. propanoic acid, methylamine, and high heat d. propene, H2SO4, H2O, and high heat e. methanoic acid, propylamine, and high heatarrow_forward5. What products would result from the following processes? Write an equation for each reaction. a. 2-Methyl-2-butanol is subjected to controlled oxidation. b. 1-Propanol is heated to 140°C in the presence of sulfuric acid. c. 3-Pentanol is subjected to controlled oxidation. d. 3-Pentanol is heated to 180°C in the presence of sulfuric acid. e. 1-Hexanol is subjected to an excess of oxidizing agentarrow_forward
- What is the major organic product obtained from the following reaction? A. 3-hydroxypropanoic acid B. 3-oxopropanoic acid C. propanedioic acid D. 1,3-propanediolarrow_forwardII- Chemical Reactions of Aldehydes and Ketones. 1. Draw the structural formula of the organic product when each of the following aldehydes is oxidized to carboxylic acid. a) Ethanal b) Pentanal c) Formaldehydes d) 3,4-dichlorohexanal 2. What are the characteristics of a positive Tollens test for aldehydes? 3. What is the oxidizing agent in Tollens solution? 4. What are the characteristics of a positive Benedict's test for aldehydes? 5. What is the oxidizing agent in benedict's solution?arrow_forwardEsters are found in some medic ines b. often fragrant essential oils in fruits and flower 41. d. found in some types of flavorings all of the above e. c.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning

Chemistry for Today: General, Organic, and Bioche...
Chemistry
ISBN:9781305960060
Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. Hansen
Publisher:Cengage Learning
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY