
Concept explainers
(a)
Interpretation:
The name of the given ketone is to be determined.
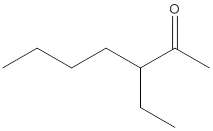
Concept Introduction:
While naming the
(b)
Interpretation:
The name of the given ketone is to be determined.
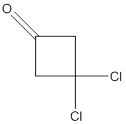
Concept Introduction:
While naming the ketones as per the IUPAC nomenclature, the naming of the compounds is done by adding a suffix-one in the end of the name. Firstly, one will find the longest chain that contains the -C=O group and then change the -e ending of the parent alkane chain to -one suffix. Then, the numbering of the chain or the ring is done in such a way that the carbonyl group is given the lowest possible number. Thereafter, apply all other rules of nomenclature as usual.
(c)
Interpretation:
The name of the given ketone is to be determined.
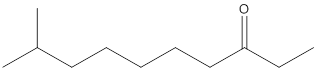
Concept Introduction:
While naming the ketones as per the IUPAC nomenclature, the naming of the compounds is done by adding a suffix-one in the end of the name. Firstly, one will find the longest chain that contains the -C=O group and then change the -e ending of the parent alkane chain to -one suffix. Then, the numbering of the chain or the ring is done in such a way that the carbonyl group is given the lowest possible number. Thereafter, apply all other rules of nomenclature as usual.
(d)
Interpretation:
The name of the given ketone is to be determined.
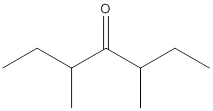
Concept Introduction:
While naming the ketones as per the IUPAC nomenclature, the naming of the compounds is done by adding a suffix-one in the end of the name. Firstly, one will find the longest chain that contains the -C=O group and then change the -e ending of the parent alkane chain to -one suffix. Then, the numbering of the chain or the ring is done in such a way that the carbonyl group is given the lowest possible number. Thereafter, apply all other rules of nomenclature as usual.
(e)
Interpretation:
The name of the given ketone is to be determined.
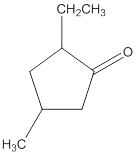
Concept Introduction:
While naming the ketones as per the IUPAC nomenclature, the naming of the compounds is done by adding a suffix-one in the end of the name. Firstly, one will find the longest chain that contains the -C=O group and then change the -e ending of the parent alkane chain to -one suffix. Then, the numbering of the chain or the ring is done in such a way that the carbonyl group is given the lowest possible number. Thereafter, apply all other rules of nomenclature as usual.
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
General, Organic, and Biological Chemistry - 4th edition
- 1. Differentiate Ethyl alcohol from methyl alcohol. 2. Compare aldehydes and ketones as to (Use acetaldehyde and acetone as examples). A. Reaction with cone. NaOH, heated B. Reaction with Tollen’s reagentarrow_forwardAn acid catalyst in nucleophilic addition of aldehydes and ketones is used for: Select one: a. Protonation of carbonyl carbon b. Making the aldehyde and ketone more susceptible to nucleophiles c. Increasing the nucleophilicity of the nucleophile d. To provide a medium for the reaction.arrow_forwardWhich of the following area. hemiacetals? b. acetals? c. hydrates?arrow_forward
- Draw the following: 1. a. 3-methyl-1-butanethiol (skunk scent) b. triphenylmethanol c. 4-(bromomethyl)-3-octanol d. 3-cyclopentenethiolarrow_forwardWhich of the following are a. hemiacetals? b. acetals? c. hydrates?arrow_forward10.38 Give the IUPAC name for each compound. a. b. C. d. e. OH f. OHarrow_forward
- 69. The structure of the aldehyde or ketone that the following compound came from: H A. III only B. IV only C. I and IV D. II only IVarrow_forwardare acetals and ketals stable to acid or base? Why?arrow_forwardGlycerol contains: a. oxygens which are each bonded to two alkyl groups b. oxygens single-bonded to primary and secondary carbons c. Oxygens double-bonded to carbon, with alkyls on both sides d. Oxygens double-bonded to carbon, with alkyls on one side only e. Oxygens double-bonded to carbon, with an alkyl on one side and an --OH on the other sidearrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning




