
Chemistry In Context
9th Edition
ISBN: 9781259638145
Author: Fahlman, Bradley D., Purvis-roberts, Kathleen, Kirk, John S., Bentley, Anne K., Daubenmire, Patrick L., ELLIS, Jamie P., Mury, Michael T., American Chemical Society
Publisher: Mcgraw-hill Education,
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 13, Problem 11Q
Here is the structural formula for the base thymine, as attached to the DNA chain.
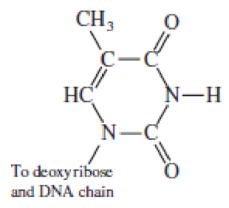
- a. Label the H atoms that can hydrogen bond with a water molecule.
- b. Use electronegativity differences to explain why only these H atoms can form hydrogen bonds.
- c. Which atoms would form hydrogen bonds with water or other
nucleic acids ?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Which one of the following is true of a molecule that can be written as multiple resonance structures?
A. the molecule must be adenine
B. the molecule must have a net positive charge
C. the molecule is unable to form H-bonds
D. the molecule has lower stability than a molecule that lacks resonance
E. the lengths of the affected bonds fall between the lengths of pure single and pure double bonds
Directions: Choose the letter of the best answer. Use a separate sheet of paper.
1.
Nutritional chemists have found that burning 1 gram of fat releases twice
the amount of heat energy as burning 1 gram of starch. Based on this
information, which type of biomolecule would cause a person to gain more weight?
A. carbohydrate
B. fat
C. proteins
D. nucleic acid
Which biomolecular group carries and passes on the hereditary information
2.
of the organism?
C. nucleic acids
D. proteins
A. carbohydrates
B. lipids
Lipids are insoluble in water because lipid molecules are
A. hydrophilic
B. neutral
Atom is the basic unit of a chemical element. What are the atoms that
3.
C. hydrophobic
D. Zwitter ions
4.
make up carbohydrates?
A. C and H
C. C, H and O
D. C, H, O and N
B. C, H and N
5. Which organic molecules are the primary energy source of the human body?
C. nucleic acids
A. carbohydrates
B. lipids
D. proteins
Watson and Crick determined that the DNA molecule has two helical strands. Each strand is made up of , which consist of a base, a deoxyribose, and a phosphate group linked together. What word most correctly fills in the blank in the preceding statement?
A.amino acids
b.monosaccharides
c.fatty acids
d.nucleotides
Chapter 13 Solutions
Chemistry In Context
Ch. 13.2 - Skill Building Small, but Important Differences...Ch. 13.2 - Prob. 13.2YTCh. 13.2 - Prob. 13.3YTCh. 13.2 - Prob. 13.4YTCh. 13.3 - Skill Building Complementary Base Sequences...Ch. 13.3 - Prob. 13.6YTCh. 13.3 - Prob. 13.7YTCh. 13.3 - Prob. 13.8YTCh. 13.4 - Prob. 13.9YTCh. 13.4 - Prob. 13.10YT
Ch. 13.5 - Scientific Practices How Is Hamburger Like Nylon?...Ch. 13.5 - Prob. 13.12YTCh. 13.5 - Scientific Practices Function Follows Form In...Ch. 13.6 - Prob. 13.14YTCh. 13 - The theme of this chapter is that DNA guides the...Ch. 13 - Prob. 2QCh. 13 - Prob. 3QCh. 13 - Consider the structural formulas in Figure 13.1....Ch. 13 - Prob. 5QCh. 13 - Prob. 6QCh. 13 - Prob. 7QCh. 13 - Prob. 8QCh. 13 - Prob. 9QCh. 13 - a. What does each letter in DNA stand for? b....Ch. 13 - Here is the structural formula for the base...Ch. 13 - Prob. 12QCh. 13 - Prob. 13QCh. 13 - Prob. 14QCh. 13 - Prob. 15QCh. 13 - Polar amino acids can be classified as acidic,...Ch. 13 - Prob. 17QCh. 13 - Prob. 18QCh. 13 - Explain one similarity and one difference between...Ch. 13 - Prob. 20QCh. 13 - Diagram the steps to produce insulin from a cow or...Ch. 13 - Prob. 22QCh. 13 - Prob. 23QCh. 13 - Prob. 24QCh. 13 - Prob. 25QCh. 13 - Prob. 26QCh. 13 - Prob. 27QCh. 13 - Prob. 28QCh. 13 - Almost all organisms use the same four bases and...Ch. 13 - Prob. 30QCh. 13 - Consider the idea of mixing genes as an...Ch. 13 - Prob. 32QCh. 13 - Prob. 33QCh. 13 - Prob. 34QCh. 13 - List two advantages and two disadvantages of...Ch. 13 - Prob. 37QCh. 13 - Prob. 38QCh. 13 - Prob. 39QCh. 13 - Recently developed techniques have dramatically...Ch. 13 - Find a transgenic organism not discussed in the...Ch. 13 - You are the head of a government facing another...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 4. Which is true about Nucleic acids: I. The polynucleotide strands of DNA are antiparallel to each other. II. The bond between base pairs are London Dispersion forces. III. In DNA, Adenine is the complementary base of Thymine. IV. The terminal Phosphate group is found at the 5’ end of the segment. A.I, II, III B.II, III, IV C.I, III, IV D.I, II, IV E.All of thesearrow_forwardHistones are A. positively charged nucleic acids. B. negative charged amino acids. C. negatively charged nucleic acids. O D. positively charged sugars. O E. positively charged amino acids.arrow_forward3 amino acids joined together by two peptide bonds.a. Dipeptideb. Tripeptidec. Tetrapeptided. Polypeptide An amino acid that can form disulfide bondsa. Cysteineb. Methioninec. Cystined. Serinearrow_forward
- What element atoms are protein made of? Unlike carbohydrates and lipids, protein contain which element? What types of food can you find protein in? What is the monomer of a protein? Draw the monomer of a proteinarrow_forwardNucleotides are molecules that make up the structures of RNA and DNA. Modify the compounds to create adenosine monophosphate (AMP).arrow_forward1. Which of the statements below best describes the following structure? a. This is the structure of testosterone. b. This is the structures of the backbone of all steroids. c. This is the structure of the backbone of all lipids. d. This is cholesterol.arrow_forward
- 6. Kevlar is a type of polymer which has a great resistance to tear. Its application includes its use in making bulletproof vests and radial tires. The two monomers used to make Kevlar are the following: H H C-0-H H-N- -N-H Monomer One Monomer Twoarrow_forwardThe "melting point" of a DNA molecule is the temperature at which the double-helical strand breaks apart. Suppose you are given two DNA samples. One sample, sample A, contains 45% G-C base pairs, while the other sample, sample B, contains 64% G-C base pairs. The total number of bases is the same in each sample. Which of the two samples, sample A or sample B, has a higher melting point? A. sample A B. sample B C. The melting point is the same for both.arrow_forwardOf the benzene molecule it is true that: a. c-c bonds have different bond lengths since they are single and double bonds. b.carries out aromatic electrophilic addition reactions c. the actual structure is a resonance hybrid d.it can be correctly represented with an equilibrium between two resonant structures.arrow_forward
- A fatty acid is a. They are naturally occurring dicarboxylic acid. b. Their carbon chains always contain at least two double bonds. c. They almost always contain an odd number of carbon atoms. O A. All three statements are true. B. Two of the three statements are true. O c. Only one of the statements is true. OD. None of the statements is true. 53arrow_forwardWhich is true about Nucleic acids: The polynucleotide strands of DNA are antiparallel to each other. The bond between base pairs are London Dispersion forces. III. In DNA, Adenine is the complementary base of Thymine. IV. The terminal Phosphate group is found at the 5’ end of the segment.arrow_forwardthe analysis of a mixture of hydrocarbon cracking products, all of which are able to be distilled, but distillation is unable to separate them cleanly. A. a pharmaceutical product containing 2 stereoisomers of the active ingredient, both of which are fairly polar, aromatic, water soluble, and decompose rather than boil. B. C. The separation of a mixture of water-soluble globular proteins of various sizes. The analysis of a mixture of reactor gases that includes methane, hydrogen, carbon dioxide, carbon monoxide, and nitrogen. D.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div
World of ChemistryChemistryISBN:9780618562763Author:Steven S. ZumdahlPublisher:Houghton Mifflin College Div World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

World of Chemistry
Chemistry
ISBN:9780618562763
Author:Steven S. Zumdahl
Publisher:Houghton Mifflin College Div

World of Chemistry, 3rd edition
Chemistry
ISBN:9781133109655
Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:Brooks / Cole / Cengage Learning

Introductory Chemistry: An Active Learning Approa...
Chemistry
ISBN:9781305079250
Author:Mark S. Cracolice, Ed Peters
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning
Biomolecules - Protein - Amino acids; Author: Tutorials Point (India) Ltd.;https://www.youtube.com/watch?v=ySNVPDHJ0ek;License: Standard YouTube License, CC-BY