
Essential Organic Chemistry (3rd Edition)
3rd Edition
ISBN: 9780321937711
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Question
Chapter 11.8, Problem 17P
Interpretation Introduction
Interpretation:
To write the mechanism for the acid-catalyzed transesterification of ethyl acetate and methanol.
Concept introduction:
Transesterification is the process of formation of a new ester molecule from the reaction of alcohol and an ester. This is like hydrolysis of ester but here nucleophile is alcohol molecule instead of
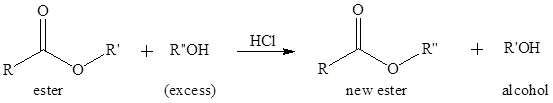
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Describe a reaction mechanism that occurs to maleic acid when hydrochloric acid is added.
Two reactions occur when sodium hydroxide is added to methyl salicylate. One is immediate and one only occurs with
reflux over time. What type of reaction occurs immediately and with which functional group on methyl salicylate does it
react? What type of reaction occurs with reflux over time and with which functional group on methyl salicylate does it
react?
Draw structural formulas for the products of the following ozonolysis reactions and name the new functional groups formed in each oxidation.
Chapter 11 Solutions
Essential Organic Chemistry (3rd Edition)
Ch. 11.1 - The aromas of many flowers and fruits are due to...Ch. 11.1 - Name the following compounds:Ch. 11.1 - Prob. 3PCh. 11.2 - Prob. 4PCh. 11.2 - Prob. 5PCh. 11.4 - a. What is the product of the reaction of acetyl...Ch. 11.4 - Prob. 7PCh. 11.5 - Using the pKa values listed in Table 11.1, predict...Ch. 11.6 - Starting with acetyl chloride, what neutral...Ch. 11.6 - Prob. 10P
Ch. 11.7 - Prob. 11PCh. 11.8 - Prob. 13PCh. 11.8 - Using the mechanism for the acidcatalyzed...Ch. 11.8 - Prob. 15PCh. 11.8 - Prob. 16PCh. 11.8 - Prob. 17PCh. 11.9 - Prob. 18PCh. 11.10 - Show how each of the following esters could be...Ch. 11.11 - Which of the following reactions would lead to the...Ch. 11.12 - Prob. 22PCh. 11.12 - Prob. 23PCh. 11.13 - Prob. 24PCh. 11.13 - Prob. 25PCh. 11.14 - Prob. 26PCh. 11.14 - Prob. 27PCh. 11.14 - Prob. 28PCh. 11.15 - Prob. 29PCh. 11.15 - How would you synthesize the following compounds...Ch. 11 - Write a structure for each of the following a. N,N...Ch. 11 - Prob. 32PCh. 11 - Which ester is more reactive, methyl acetate or...Ch. 11 - What products would be formed from the reaction of...Ch. 11 - What products would be obtained from the following...Ch. 11 - Prob. 36PCh. 11 - a. Which compound would you expect to have a...Ch. 11 - a. List the following esters in order of...Ch. 11 - D. N. Kursanov, a Russian chemist, proved that the...Ch. 11 - Prob. 40PCh. 11 - Using an alcohol for one method and an alkyl...Ch. 11 - Prob. 42PCh. 11 - Prob. 44PCh. 11 - Prob. 45PCh. 11 - Prob. 46PCh. 11 - Prob. 47PCh. 11 - Prob. 48PCh. 11 - Prob. 49PCh. 11 - Show how the following compounds could be prepared...Ch. 11 - Prob. 51PCh. 11 - Prob. 52PCh. 11 - Prob. 53P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Write chemical reaction of the epoxide preparation by reacting cis-2-pentene with a peroxycarboxylic acid. peroxycarboxylic acidarrow_forwardGive the reagents and intermediate products for the following two-step reaction.arrow_forwardSYNTHESIS OF ESTERS VIA NUCLEOPHILIC ACYL SUBSTITUTION Write the chemical equation involved in the reaction between the excess acid and NaHCO3. Explain why NaHCO3 is preferred over NaOH for the neutralization of excess acid. How was excess alcohol eliminated from the crude product.arrow_forward
- in the oxidation of cyclohexanol to cyclohexanone, what purpose does the acetic acid serve?arrow_forwardExplain why methyl trifluoroacetate, CF3CO2CH3, is more reactive than methyl acetate, CH3CO2CH3, in nucleophilic acyl substitution reactions.arrow_forwardDraw out the reaction mechanism for cyclohexanol to cyclohexanone. Sodium hypochlorite oxidation of an alcohol to a ketone with the product being cyclohexanone.arrow_forward
- Draw and explain the mechanism for the ammoxidation of propene which leads to the formation of acrolein.arrow_forwardDescribe the reaction where benzil is prepared by the oxidation of an a-hydroxyketone, benzoin and uses nitric acid to preform oxidation.arrow_forwardOn synthesis of esters via nucleophilic acyl substitution: Write the chemical equation involved in the reaction between the excess acid and NaHCO3. Given this, briefly explain why NaHCO3 is preferred over NaOH for the neutralization of excess acid. Also, explain how excess alcohol was eliminated from the crude product.arrow_forward
- Provide the product for the following transformation:arrow_forwardWhen 4-hydroxybutanoic acid is treated with an acid catalyst, it forms a lactone (a cyclic ester). Draw the structural formula of this lactone and propose a mechanism for its formationarrow_forwardDuring the decarboxylation of acetoacetate, what is the nucleophile and what is the electrophile? Is the formation of a schiff base simply a preparation step or is the formation of a schiff base formed also by nucleophile/electrophile interaction?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning

Macroscale and Microscale Organic Experiments
Chemistry
ISBN:9781305577190
Author:Kenneth L. Williamson, Katherine M. Masters
Publisher:Brooks Cole

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:9781285869759
Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:Cengage Learning
Seven Name Reactions in One - Palladium Catalysed Reaction (047 - 053); Author: Rasayan Academy - Jagriti Sharma;https://www.youtube.com/watch?v=5HEKTpDFkqI;License: Standard YouTube License, CC-BY