
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 8.5, Problem 8.27P
On a separate piece of paper, draw a mechanism for the transformation in the previous problem (8.26).
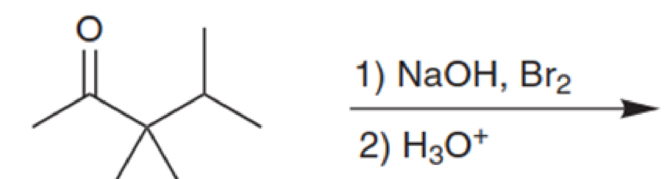
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
7.64 Fluticasone, the chapter-opening molecule, can be prepared by the following reaction. Draw
a stepwise mechanism for this reaction.
F
SH
HO
HO
BRCH,F
K2CO3
DMF
H
A
fluticasone
How does each of the following changes affect the rate of an E2 reaction? (a, b, and c)
Draw the structures of 2 anionic groups that are not considered good leaving groups in an SN2
reaction:
6.
(4 pts) Which of the following would best accomplish a SN2 reaction as a nucleophile with a primary
Chapter 8 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.2 - Prob. 8.9PCh. 8.2 - Prob. 8.10PCh. 8.2 - Prob. 8.11PCh. 8.2 - Prob. 8.12P
Ch. 8.2 - Prob. 8.13PCh. 8.3 - Predict the products of each of the following...Ch. 8.3 - Predict the products of each of the following...Ch. 8.3 - Predict the products of each of the following...Ch. 8.3 - Predict the products of each of the following...Ch. 8.4 - Prob. 8.20PCh. 8.4 - Prob. 8.21PCh. 8.4 - Prob. 8.22PCh. 8.4 - Prob. 8.23PCh. 8.5 - Prob. 8.25PCh. 8.5 - Prob. 8.26PCh. 8.5 - On a separate piece of paper, draw a mechanism for...Ch. 8.6 - Prob. 8.29PCh. 8.6 - Predict the major product for each of the...Ch. 8.6 - Predict the major product for each of the...Ch. 8.6 - Predict the major product for each of the...Ch. 8.6 - Identify the reagents you would use to achieve...Ch. 8.6 - Identify the reagents you would use to achieve...Ch. 8.6 - Identify the reagents you would use to achieve...Ch. 8.7 - Predict the major product for each of the...Ch. 8.7 - Predict the major product for each of the...Ch. 8.7 - Predict the major product for each of the...Ch. 8.7 - Prob. 8.42PCh. 8.7 - Prob. 8.43PCh. 8.7 - Prob. 8.44PCh. 8.7 - Prob. 8.45PCh. 8.7 - Prob. 8.47PCh. 8.7 - Prob. 8.48PCh. 8.7 - Prob. 8.49PCh. 8.7 - Prob. 8.50PCh. 8.8 - Prob. 8.52PCh. 8.8 - Prob. 8.53PCh. 8.8 - Prob. 8.54PCh. 8.8 - Prob. 8.55PCh. 8.8 - Prob. 8.57PCh. 8.8 - Prob. 8.58PCh. 8.8 - Prob. 8.59PCh. 8.8 - Prob. 8.60PCh. 8.8 - Propose a mechanism for each of the following...Ch. 8.8 - Propose a mechanism for each of the following...Ch. 8.8 - Prob. 8.64PCh. 8.9 - Prob. 8.66PCh. 8.9 - Prob. 8.67PCh. 8.9 - Prob. 8.68PCh. 8.9 - Prob. 8.69PCh. 8.9 - Prob. 8.70PCh. 8.9 - Prob. 8.71PCh. 8.9 - Prob. 8.72PCh. 8.9 - Identify what reagents you would use to achieve...Ch. 8.9 - Identify what reagents you would use to achieve...Ch. 8.9 - Identify what reagents you would use to achieve...Ch. 8.10 - Prob. 8.78PCh. 8.10 - Prob. 8.79PCh. 8.10 - Prob. 8.80PCh. 8.10 - Propose a synthesis for each of the following...Ch. 8.10 - Propose a synthesis for each of the following...Ch. 8.10 - Propose a synthesis for each of the following...Ch. 8.10 - Propose a synthesis for each of the following...
Additional Science Textbook Solutions
Find more solutions based on key concepts
Practice Problem 1.25
What do the bond angles of boron trifluoride suggest about the hybridization state of the...
Organic Chemistry
For Practice 1.1
Is each change physical or chemical? Which kind of property (chemical or physical) is demonst...
Principles of Chemistry: A Molecular Approach (3rd Edition)
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
a. How can aspirin be synthesized from benzene? b. Ibuprofen is the active ingredient in pain relievers such as...
Organic Chemistry (8th Edition)
Draw the enol tautomers for each of the following compounds. For compounds that have more than one enol tautome...
Organic Chemistry
What is the osmotic pressure in torr of a 0.010 M aqueous solution of a molecular compound at 25C?
Chemistry: The Molecular Nature of Matter
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 7.61 Draw a stepwise mechanism for the following reaction that illustrates how two substitution products are formed. Explain why 1-bromohex-2- ene reacts rapidly with a weak nucleophile (CH3OH) under SN1 reaction conditions, even though it is a 1 ° alkyl halide. OCH3 CH;OH Br OCH3 + HBr 1-bromohex-2-enearrow_forwardc) Show how you would carry out the following transformations. (4 Marks) OHarrow_forwardb) When the compound below is treated with a base, kinetic product A and thermodynamic product B, can be formed depending on the base and temperature. Draw the structures for A and B and write the mechanism for the formation of the thermodynamic product (6 Marks) Thermodynamic product Base A Br Kinetic Productarrow_forward
- 9.49 Draw a stepwise mechanism for the following reaction. он H,SO4 H20arrow_forward1. Please name the following compounds. (9 points) Br CI Br Br 2. Please draw the major product(s) of the following reaction and show the mechanism. Is there a reason why one stereochemistry is preferred over the other for this reaction? (9 points)arrow_forward7.64 In each of the following cases, draw the structure of an alkyl halide that will undergo an E2 elimination to yield only the indicated alkene. (a) (c) ? E2 ? £2, lifw (b) (d) ? E2 agent (esion 103onia Die to ? E2 otoros slog s to seu si b)arrow_forward
- 7.68 When a single compound contains both a nucleophile and a leaving group, an intramolecular reaction may occur. With this in mind, draw the product of the following reaction. LOH -OH C,H10O2 + Br 'Br Br H,0arrow_forward4) Using the Wieland-Miescher ketone as your starting material, how would you synthesize the desired product below? Note: You do not need to show mechanisms for each reaction, only conditions. (8 points) OH Me Mell ? HO H Wieland-Miescher Ketonearrow_forward(2) Draw the arrow-pushing mechanism of the following reaction: (10 pts) 1) AlCl; CI 2) H2Oarrow_forward
- 7.13 A proton (H+) from trifluoromethanesulfonic acid, CF3SO2OH, can add to the alkyne shown here to yield two different carbocation products. (a) Draw the mechanism for each of these steps, along with the corresponding products. (b) Which carbocation is more stable? H3C H₂ IU C-C=CH + H-OSO₂CF3 H₂ ?arrow_forwardFill in the product, starting material, or reagent for each reaction. "No reaction" is a possibility. Also, be sure to show stereochemistry and anyrearrangements if they occur. 6. 1) NANH2 2) CH;CH,Br 3) Lindlar's H2 H3C=arrow_forward9.45 Draw the expected product of each of the following reactions, showing stereochemistry where appropriate: me ? ? 9.46 Compound A is an alkyne that reacts with two equivalents of H₂ in the presence of Pd to give 2,4,6-trimethyloctane. Maho NH3 Draw the structure of compound A. How many chiral centers are present in compound A? Identify the locants for the methyl groups in compound A. Explain why the locants are not 2, 4, and 6 as seen in the product of hydrogenation.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
GCSE Chemistry - Differences Between Compounds, Molecules & Mixtures #3; Author: Cognito;https://www.youtube.com/watch?v=jBDr0mHyc5M;License: Standard YouTube License, CC-BY