
General, Organic, and Biological Chemistry - 4th edition
4th Edition
ISBN: 9781259883989
Author: by Janice Smith
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8.3, Problem 8.3PP
Which compounds are water soluble?
a.
b.
c. 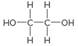
d.
e.
More Practice: Try Problems 8.33, 8.37, 8.38.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
10:13
Formula
AgCl
LaF3
CuBr
CaF₂
Which of the following salts would have the
lowest solubility in water?
A) AgCl
B) LaF3
C) CuBr
Question 1 of 16
D) CaF₂
Ksp
1.8 x 10-1⁰
2 x 10-1⁹
5.3 x 10-⁹
3.5 x 10-¹1
Submit
Periodic Table
1 Learning Objective: 8.F Calculate the concentration of ions in solutions and classify electrolytes by strength.
Adaptive Assignment
e See page 345
Question
1st attempt
See Periodic Table O See Hint
The concentration of Mg2+ in a sample of coastal seawater is 1.08 g/kg. What is the molar concentration of Mg2* in water with a density
of 1.02 g/mL?
M
A SUBMIT ANSWER
MacBook Air
DO
esc
FO
%23
24
96
2.
3
5
7
T
Y
OP
F
H.
K
C
V
M
DI
S'
Boost
3:27 PM
O 100% Ca
Done
education.wiley.com AA C
1.14 Solubility
Based on the strength and type of
intermolecular forces, one would expect
neopentane (shown below) to be
in water.
H-C-H
H
H.
H-C-
-C-
C-H
H
H
H-C-H
Done
soluble
not soluble
Chapter 8 Solutions
General, Organic, and Biological Chemistry - 4th edition
Ch. 8.1 - Classify each substance as a heterogeneous...Ch. 8.1 - Use the appearance of each product to classify it...Ch. 8.2 - Consider the following diagrams for an aqueous...Ch. 8.2 - Classify each solution as an electrolyte or...Ch. 8.2 - Using the given number of moles, determine how...Ch. 8.2 - Prob. 8.2PPCh. 8.2 - A solution contains the following ions:...Ch. 8.2 - If a solution contains 125 mEq of Na+ per liter,...Ch. 8.3 - Which compounds are water soluble? a. NaNO3 b. CH4...Ch. 8.3 - Prob. 8.7P
Ch. 8.3 - Use the solubility rules to predict whether the...Ch. 8.3 - Use the solubility rules for ionic compounds to...Ch. 8.4 - Why does a soft drink become "flat" faster when it...Ch. 8.4 - Predict the effect each change has on the...Ch. 8.5 - A commercial mouthwash contains 4.3 g of ethanol...Ch. 8.5 - What is the weight/volume percent concentration of...Ch. 8.5 - Prob. 8.6PPCh. 8.5 - Prob. 8.7PPCh. 8.5 - A drink sold in a health food store contains 0.50%...Ch. 8.5 - Prob. 8.12PCh. 8.5 - What is the concentration in parts per million of...Ch. 8.6 - Prob. 8.10PPCh. 8.6 - Prob. 8.13PCh. 8.6 - Prob. 8.11PPCh. 8.6 - Prob. 8.12PPCh. 8.6 - How many grams of NaCl are contained in each of...Ch. 8.6 - How many milliliters of a 0.25 M sucrose solution...Ch. 8.7 - What is the concentration of a solution formed by...Ch. 8.7 - If the solution of A+B- in X is diluted, which...Ch. 8.7 - Prob. 8.15PPCh. 8.7 - Prob. 8.16PCh. 8.8 - What is the boiling point of a solution prepared...Ch. 8.8 - Representations A, B, and C each show an aqueous...Ch. 8.8 - Prob. 8.18PPCh. 8.8 - What is the melting point of a solution that is...Ch. 8.9 - Which solution in each pair exerts the greater...Ch. 8.9 - Prob. 8.19PCh. 8.9 - Consider the two aqueous solutions separated by a...Ch. 8.9 - What happens to a red blood cell when it is placed...Ch. 8 - Prob. 21PCh. 8 - Prob. 22PCh. 8 - Prob. 23PCh. 8 - Which representation of molecular art better shows...Ch. 8 - Classify each of the following as a solution,...Ch. 8 - Classify each of the following as a solution,...Ch. 8 - Prob. 27PCh. 8 - Label each diagram as a strong electrolyte, weak...Ch. 8 - Prob. 29PCh. 8 - Prob. 30PCh. 8 - Prob. 31PCh. 8 - Prob. 32PCh. 8 - Consider a mixture of two substances shown in blue...Ch. 8 - Which diagram (C or D) best represents what occurs...Ch. 8 - If the solubilityofKClin 100 mL of H2O is 34 g at...Ch. 8 - If the solubilityofsucrosein 100 mL of H2O is 204...Ch. 8 - Prob. 37PCh. 8 - Prob. 38PCh. 8 - Using the ball-and-stick model for methanol...Ch. 8 - Prob. 40PCh. 8 - Prob. 41PCh. 8 - Prob. 42PCh. 8 - Prob. 43PCh. 8 - Prob. 44PCh. 8 - Prob. 45PCh. 8 - Prob. 46PCh. 8 - Prob. 47PCh. 8 - How is the solubility of helium gas in water...Ch. 8 - Use the solubility rules listed in Section 8.3B to...Ch. 8 - Use the solubility rules listed in Section 8.3B to...Ch. 8 - Prob. 51PCh. 8 - Prob. 52PCh. 8 - Prob. 53PCh. 8 - Prob. 54PCh. 8 - Prob. 55PCh. 8 - Prob. 56PCh. 8 - Prob. 57PCh. 8 - Prob. 58PCh. 8 - How would you use a 250-mL volumetric flask to...Ch. 8 - How would you use a 250-mLvolumetric flask to...Ch. 8 - Prob. 61PCh. 8 - Prob. 62PCh. 8 - Prob. 63PCh. 8 - Prob. 64PCh. 8 - Prob. 65PCh. 8 - What is the molarity of a 20.0% (v/v) aqueous...Ch. 8 - Prob. 67PCh. 8 - Prob. 68PCh. 8 - Prob. 69PCh. 8 - Prob. 70PCh. 8 - Prob. 71PCh. 8 - Prob. 72PCh. 8 - Prob. 73PCh. 8 - Prob. 74PCh. 8 - Prob. 75PCh. 8 - Prob. 76PCh. 8 - Prob. 77PCh. 8 - Representations A (containing 1.0 mol ofNaCl) and...Ch. 8 - What is the boiling point of a solution that...Ch. 8 - Prob. 80PCh. 8 - If 150 g of ethylene glycol (C2H6O2) is added to...Ch. 8 - Prob. 82PCh. 8 - Prob. 83PCh. 8 - Prob. 84PCh. 8 - Which solution in each pair has the higher melting...Ch. 8 - Prob. 86PCh. 8 - A flask contains two compartments (A and B) with...Ch. 8 - A flask contains two compartments (A and B) with...Ch. 8 - The molecular art illustrates a red blood cell in...Ch. 8 - Prob. 90PCh. 8 - Prob. 91PCh. 8 - Explain why more sugar dissolves in a cup of hot...Ch. 8 - If the concentration of glucose in the blood is...Ch. 8 - Prob. 94PCh. 8 - Mannitol, a carbohydrate, is supplied as a 25%...Ch. 8 - A patient receives 750 ml, of a 10.% (w/v) aqueous...Ch. 8 - Explain why a cucumber placed in a concentrated...Ch. 8 - Explain why a cucumber placed in a concentrated...Ch. 8 - Prob. 99PCh. 8 - Prob. 100PCh. 8 - Prob. 101PCh. 8 - Prob. 102PCh. 8 - The therapeutic concentration—the concentration...Ch. 8 - Prob. 104CP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- ⒸMacmillan Learning What is the concentration of a solution made by diluting 45 mL of 6.0 M HCl to a final volume of 750 mL? 80 F3 concentration: $ 4 288 F4 % 5 T F5 ^ 6 MacBook Air F6 & 7 - F7 11 00* 8 DII F8 ( 9 DD F9 O ) O. O F10 P 3) F11 { + r = F12 M deletearrow_forwardMacmillan Learning What is the concentration of a solution made by diluting 25 mL of 6.0 M HCl to a final volume of 750 mL? concentration: Marrow_forwardQuestion 6 In the following reaction, 2HCI(aq) + CaCO3(s) CO28) + H20(1) + CaCl2(aq) What is the molarity of 500mL of the CaCl2 produced, if 70mL of 1.5M HCI was used? TTTF Paragraph Arial v 3 (12pt) Mashups HTHL CSS Path: p MAY tv N A の Aa 11 6621715 DD 20 000 D00 F10 F9 F6 F7 FB F3 F4 F5 24 & 4 7 8 R Y この < co トarrow_forward
- Describe the preparation of a 780 mL of 0.0160 M AgNO, from the solid reagent. mL. Dissolve g AgNO, in enough water to give a final volume of Submit b 3.20 L of 0.203 M HCI, starting with a 6.00 M solution of the reagent. Take mL of the 6.00 M HCl and dilute to L with water. C 780 mL of a solution that is 0.0160 M in K*, starting with solid K. Fe (CN), Dissolve g KA Fe (CN), in enough water to give a final volume of mL. d780 mL of 1.10% (w/v) aqueous BaCl2 from a 0.400 M BaCl2 solution. Take mL of the 0.400 M BaCl2 solution and dilute to mL with water. e 7.60 L of 0.100 M HCIO4 from the commercial reagent [71.0% HCIO4 (w w), sp gr 1.67]. Take mL of the concentrated reagent and dilute to L with water.arrow_forwardr 10 Homework i 3 ed ok t t nces Select the products formed when the two solutions given are mixed together. Ca(CIO4)2(aq) + Na2CO3(aq) → ? Check All That Apply CaCO3(s) Na2(CIO4)2(s) NaCIO4(aq) Saved Ca2CO3(aq) 10 C Noxt .edu%252Fwebaparrow_forwardQuestion 24 Choose the best answer for the pOH of a solution containing 4.0 x 10- M of [H]. 6.3 x 10-6 2.5 x 10-10 4.4 9.6 Question 25arrow_forward
- # 3 20 E D The molar solubility of aluminum hydroxide in a water solution is Submit Answer 54 $ 888 R F F4 % 5 Retry Entire Group 9 more group attempts remaining T Cengage Learning Cengage Technical Support F5 G A 6 MacBook Air S F6 Y H & 7 V F7 U * 00 8 ▶11 Values it needed for this question. F8 1 9 K F9 O 1 0 J F10 P M. F11 =arrow_forwardExercises 1. Which solvent, water (H₂O) or hexane (C6H₁4), would you use to dissolve the following solutes? a. Octane (C8H18) b. HF C. (NH4)2SO4arrow_forwardBoost 4:24 PM O 99% Done education.wiley.com AA 4 Solubility Of the given compounds, which is more soluble in water? Lö: H2C- H- CH2 H2Č. H2 O both are equally soluble Hint Save for Laterarrow_forward
- How many mL of 0.668 M HBr are needed to dissolve 7.69 g of MgCO,? 2HB1(aq) + MgCO3(s) MgBrz(aq) + H50(1) + CO2(g) mL 9 more group attempts remaining Retry Entire Group Submit Answer hp ins prt sc dele f12 f11 トト f8 f9 f10 f6 17 L. 80arrow_forwardProcedure: 1) Preparation of 100 mL 0.1N Na2CO3 2) How much is the molarity of the prepared solution 3) Dilute the solution to get 100 ml 0.01 N. 4) How much the Molarity of the diluted solution. M.wt of Na2CO3 = 106 g mol-1 Eq.wt = 106 / 2 = 53 g eq1 %3D Every 53 g in 1000 ml How much?(x) » in 100 mL → to give 0.1 N 53 x 100 x 0.1 X = 0.53 g 1000 x 1 : weigh 0.53 g Na2CO3 place it in a beaker, add about 30-50 ml distilled water , stir well. Transfer the solution to 100 ml volumetric flask, then fill to the mark with distilled water to get 0.1 N solution. • Put your results and your answers in the following table : Wt. NażCO3 (g) Normality N eq L-1 Molarity M mol L-1 53 ? 0.53 After dilution 0.01arrow_forwardMacmillan Learning What is the concentration of a solution made by diluting 75 mL of 6.0 M HCl to a final volume of 750 mL? concentration: घ Marrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Acid-Base Titration | Acids, Bases & Alkalis | Chemistry | FuseSchool; Author: FuseSchool - Global Education;https://www.youtube.com/watch?v=yFqx6_Y6c2M;License: Standard YouTube License, CC-BY