
Essential Organic Chemistry (3rd Edition)
3rd Edition
ISBN: 9780321937711
Author: Paula Yurkanis Bruice
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 8, Problem 39P
Draw the major product obtained when each of the following
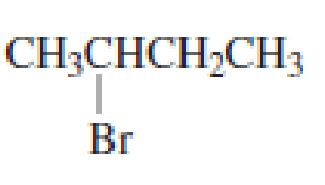
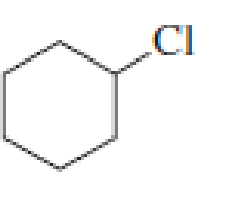
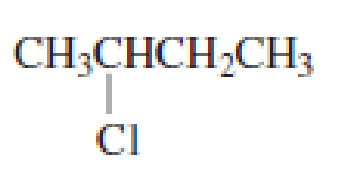
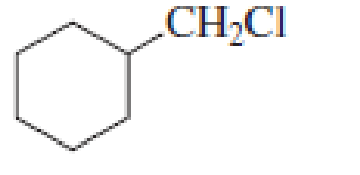
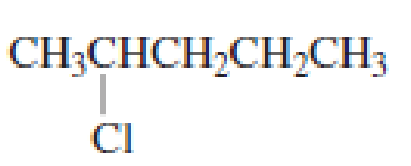
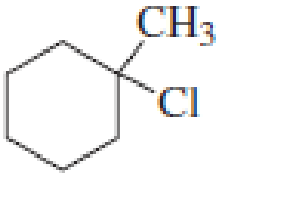
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the major product formed when the following compounds undergo an E1 reaction?
Part A
What is the major product formed when the following compound undergo an E1 reaction?
CH, CH
CH;CH,C–CHCH;CH3
Br
for bonds ond chargee) Atoms gnd Ter
What products are obtained from the reaction of each of the following alkenes with OsO4 followed by aqueous H2O2?
Chapter 8 Solutions
Essential Organic Chemistry (3rd Edition)
Ch. 8.1 - Prob. 2PCh. 8.1 - Does increasing the energy barrier for an SN2...Ch. 8.1 - Arrange the following alkyl bromides in order from...Ch. 8.2 - Prob. 7PCh. 8.2 - Which reaction in each of the following pairs...Ch. 8.2 - Prob. 9PCh. 8.2 - Prob. 11PCh. 8.3 - Draw the substitution products that will be formed...Ch. 8.4 - Arrange the following alkyl halides in order from...Ch. 8.5 - Prob. 14P
Ch. 8.5 - Which of the following reactions will go faster if...Ch. 8.6 - After a proton is removed from the OH group, which...Ch. 8.6 - Draw the product of each of the following...Ch. 8.9 - Prob. 20PCh. 8.9 - Prob. 21PCh. 8.11 - Why do the SN1/E1 reactions of tertiary alkyl...Ch. 8.11 - Prob. 23PCh. 8.11 - Prob. 24PCh. 8.12 - Prob. 25PCh. 8.12 - Prob. 26PCh. 8.12 - Prob. 27PCh. 8.12 - a In which solvent would tert-butyl bromide...Ch. 8.13 - What would be the best way to prepare the...Ch. 8 - Methoxychlor is an insecticide that was intended...Ch. 8 - Prob. 31PCh. 8 - Prob. 32PCh. 8 - Prob. 33PCh. 8 - Prob. 34PCh. 8 - Prob. 35PCh. 8 - Prob. 36PCh. 8 - Explain how the following changes would affect the...Ch. 8 - Prob. 38PCh. 8 - Draw the major product obtained when each of the...Ch. 8 - Which alkyl halide in Problem 39 can undergo an El...Ch. 8 - Prob. 42PCh. 8 - Prob. 43PCh. 8 - Prob. 44PCh. 8 - Prob. 45PCh. 8 - Starting with bromocyclohexane, how could the...Ch. 8 - Prob. 48PCh. 8 - Fill in the blanks in the following chemical...Ch. 8 - For each of the following alkyl halides, indicate...Ch. 8 - Prob. 51PCh. 8 - a. Explain why 1-bromo-2,2-dimethylpropane has...Ch. 8 - An ether can be prepared by an SN2 reaction of an...Ch. 8 - Give two sets of reactants (each set including an...Ch. 8 - Show how the following compounds could be...Ch. 8 - Prob. 56PCh. 8 - Prob. 57PCh. 8 - Draw the structures of the products obtained from...Ch. 8 - cis-4-Bromocyclohexanol and...Ch. 8 - Prob. 60PCh. 8 - Prob. 61PCh. 8 - Prob. 62PCh. 8 - Prob. 63PCh. 8 - In which solventethanol or diethyl etherwould the...Ch. 8 - The pKa of acetic acid in water is 4.76. What...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Which of the following compounds will not undergo keto-enol tautomerism? ОН OH ОН OH ОНarrow_forwardDraw the major organic product formed in each of the following transition-metal catalyzed reactions:arrow_forwardRank the following carbonyl compounds in order of increasing reactivity in nucleophilic substitution reactions: H I II III IV ZIarrow_forward
- What is the major product formed when the following compound undergoes an E1 reaction?arrow_forwardDraw the structures of the initially formed enol tautomers in the reactions of propyne and dicyclohexylethyne with dicyclohexylborane followed by NaOHNaOH and H2O2H2O2arrow_forwardWhich of the following sequences of reactions would convert toluene to 2-bromo-4-cyanotoluene? ? Br CN Nitration, bromination, reduction, diazotization, reaction with cyanide anion Bromination, nitration, reduction, diazotization, reaction with cyanide anion Bromination, nitration, diazotization, reduction , reaction with cyanide anion Nitration, bromination, diazotization, reduction, reaction with cyanide anionarrow_forward
- Determine a stepwise mechanism for the following reaction that illustrates why two substitution products are formed. Explain why 1-bromohex-2-ene reacts rapidly with a weak nucleophile (CH3OH) under Syl reaction conditions, even though it is a 1° alkyl halide. 1-bromohex-2-ene Part 1: Br CH₂OH The first step in the reaction proceeds according to which mechanism? CH₂CH₂CH₂CH=CHCH₂ + CH₂OH CH₂CH₂CH₂CH: Part 2: Br CHCH₂ Draw the missing resonance contributor. OCH3 + CH3CH₂CH₂CH=CHCH₂ CH3CH₂CH₂CH=CH-CH₂ + Br Br OCH 3 H₂CH₂CH₂CH=CH₂ view structure + + Br HBr Xarrow_forwardDraw the starting structure that would lead to the major product shown under the provided conditions. Drawing H3O+ HOarrow_forwardIdentify the dominant elimination mechanism (E1 or E2) for the following reaction. NaOH Neither Е2 Е1arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Alcohols, Ethers, and Epoxides: Crash Course Organic Chemistry #24; Author: Crash Course;https://www.youtube.com/watch?v=j04zMFwDeDU;License: Standard YouTube License, CC-BY