
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6.6B, Problem 6.9P
The light-initiated reaction of 2,3-dimethylbut-2-ene with N-bromosuccimmide (NBS) gives two products:
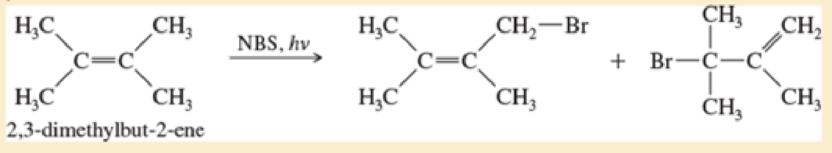
- a. Give a mechanism for this reaction, showing how the two products arise as a consequence of the resonance-stabilized intermediate.
- b. The bromination of cyclohexene using NBS gives only one major product, as shown on the previous page Explain why there is no second product from an allylic shift.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
1. Write the mechanism for the electrophilic aromatic
substitution for nitration of the following compound. Both
rings must be accounted for in their individual EAS.
2. Show an energy diagram for the reaction progress for both
rings.
-Ö:
oso
:Ö
1) a. Propose a mechanism to account for the following reaction: 1-methylcyclohex-2-enol reacts
with HI to give 3-iodo-3-methylcyclohex-1-ene and 3-iodo-1-methylcyclohex-1-ene.
b. When 1-(1-methylethyl)cyclohexene (1-isopropylcyclohexene) is treated with a solution of HBr
in Et₂0 2-bromo-2-cyclohexylpropane is the resulting product. Write the reaction and propose a
mechanism using curved arrows to indicate the electron flow in each step
c. Draw a mechanism that accounts for the following transformation.
HO.
LOCH3
com
OCH3
CF3CO₂H
You must prepare the most stable possible alkene starting from the starting material given
below and following an E2 elimination reaction.
H
Base
Ö
most stable alkene
CH3
Ph
Et
Br
Ph
Chapter 6 Solutions
Organic Chemistry (9th Edition)
Ch. 6.1 - Classify each compound as an alkyl halide, a vinyl...Ch. 6.2 - Give the structures of the following compounds. a....Ch. 6.2 - For each of the following compounds, A. give the...Ch. 6.3E - Prob. 6.4PCh. 6.4 - Prob. 6.5PCh. 6.5A - For each pair of compounds, predict which compound...Ch. 6.5B - Prob. 6.7PCh. 6.6B - Prob. 6.8PCh. 6.6B - The light-initiated reaction of...Ch. 6.6B - Show how free-radical halogenation might be used...
Ch. 6.7 - Prob. 6.11PCh. 6.7 - Prob. 6.12PCh. 6.8 - Prob. 6.13PCh. 6.9 - Predict the major products of the following...Ch. 6.9 - Prob. 6.15PCh. 6.10A - Prob. 6.16PCh. 6.11A - When diethyl ether (CH3CH2OCH2CH3) is treated with...Ch. 6.11B - Prob. 6.18PCh. 6.11B - For each pair of compounds, state which compound...Ch. 6.12 - Prob. 6.20PCh. 6.12 - Under appropriate conditions...Ch. 6.13 - Propose an SN1 mechanism for the solvolysis of...Ch. 6.13B - Prob. 6.23PCh. 6.13B - 3-Bromocyclohexene is a secondary halide, and...Ch. 6.15 - Prob. 6.25PCh. 6.15 - Prob. 6.26PCh. 6.16 - For each reaction, give the expected substitution...Ch. 6.16 - Prob. 6.28PCh. 6.16 - Prob. 6.29PCh. 6 - Prob. 6.30SPCh. 6 - Draw the structures of the following compounds. a....Ch. 6 - Give systematic (IUPAC) names for the following...Ch. 6 - Prob. 6.33SPCh. 6 - Predict the compound in each pair that will...Ch. 6 - Prob. 6.35SPCh. 6 - Give two syntheses for (CH3)2CHOCH2CH3, and...Ch. 6 - Prob. 6.37SPCh. 6 - Prob. 6.38SPCh. 6 - Chlorocyclohexane reacts with sodium cyanide...Ch. 6 - Give the substitution products expected from...Ch. 6 - Prob. 6.41SPCh. 6 - Prob. 6.42SPCh. 6 - Two of the carbocations in Problem6-42 are prone...Ch. 6 - Prob. 6.44SPCh. 6 - Predict the products of the following SN2...Ch. 6 - Prob. 6.46SPCh. 6 - Strawberry growers have used large quantities of...Ch. 6 - A solution of pure (S)-2-iodobutane ([]=+15.90) in...Ch. 6 - Prob. 6.49SPCh. 6 - Give a mechanism to explain the two products...Ch. 6 - Prob. 6.51SPCh. 6 - Because the SN1 reaction goes through a flat...Ch. 6 - Prob. 6.53SPCh. 6 - Furfuryl chloride can undergo substitution by both...Ch. 6 - Prob. 6.55SPCh. 6 - The following reaction takes place under...Ch. 6 - Propose mechanisms to account for the observed...Ch. 6 - Prob. 6.58SPCh. 6 - Prob. 6.59SP
Additional Science Textbook Solutions
Find more solutions based on key concepts
Practice Problem ATTEMPT
Write the rate expressions for each of the following reactions:
(a)
(b)
(c)
Chemistry
Real walls are never totally adiabatic. Use your experience to order the following walls in increasing order wi...
Thermodynamics, Statistical Thermodynamics, & Kinetics
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach
22.102 Write the structures of the cis and tram isomers, if any, for the following compounds:
Chemistry: The Molecular Nature of Matter
4.1 Write the symbols for the following elements.
a. copper
b. platinum
c. calcium
d. manganese
e. Iron
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- 1) a. Propose a mechanism to account for the following reaction: 1-methylcyclohex-2-enol reacts with HI to give 3-iodo-3-methylcyclohex-1-ene and 3-iodo-1-methylcyclohex-1-ene. b. When 1-(1-methylethyl)cyclohexene (1-isopropylcyclohexene) is treated with a solution of HBr in Et₂O 2-bromo-2-cyclohexylpropane is the resulting product. Write the reaction and propose a mechanism using curved arrows to indicate the electron flow in each step. c. Draw a mechanism that accounts for the following transformation. НО. LOCH3 OCH 3 CF3CO₂Harrow_forward1. What product is expected to form in an Sy2reaction of OH¯with (R)-2-bromobutane? Show the stereochemistry of the reactant and the product. 2. What product is expected to form from the SN2reaction of 1-bromopentane with а. КОН? B. Nal? 3. What product would you expect to obtain from a nucleophilic substitution reaction of (S)-2- bromohexane with CH3CO,? Assume that inversion configuration occurs, and show the stereochemistry of both reactant and product? 4. Predict whether each of the following substitution reactions is likely t Sylor SN2. CI OAc CH3CO, Na+ CH3CO2H, H20 а. CH2BR CH2OAC CH3CO2- Na+ DMF b. OH HCI CI CH3OH с. CH3 CH3 Na+ SCH3 H2C=CH2B d. H2C=CCH,SCH3 CH3CN 5. Setting aside the double bond stereochemistry, what products would you expect from elimination reactions of the following alkyl halides? CH3 ÇI CH3 CH3CHCH2-C-CHCH3 ČH3arrow_forward1. What effect would you expect on the rate of reaction of ethanol with 2-iodo-2-methyl-butane if the concentration of the halide is tripled? 2. Explain the difference between addition, elimination and substitutionreactions. 3. Optically active 2-butanol slowly racemizes on standing in dilute sulfuricacid. Propose a mechanism to account for this observation.arrow_forward
- Provide the product most likely to form from reaction of each reagent with 1-methylcyclohexene. You may ingore stereochemistry for this question. Br2 H-Br H-Br, PhC(O)OOC(O)Ph N-bromosuccinimide, PhC(O)OOC(O)Ph A. B. C. D.arrow_forwardConsider the following nucleophilc substitution. LOCH3 Br OCH3 в A a. Identify the reaction conditions A and specify the reaction mechanism for the nucleophilic substitution. Explain your choice. b. Explain why the transformation does not give rise to significant elimination. c. Which product(s) B is formed in the reaction? Please specify the stereochemistry clearly with reference to the reaction mechanism.arrow_forwardII. Alcohols in synthesis A. What are the strategies for the synthesis of alcohols by nucleophilic substitution reactions using halide leaving groups with hydroxide as a nucleophile? How do you predict possible by-products that could arise from this strategy? How do you use the less acetate nucleophile followed by treatment with NaOH to avoid by-products?arrow_forward
- Benzene undergoes electrophilic aromatic substitution reactions with aziridines in the presence of a Lewis acid such as AlCl3. a. What are the major and minor products of the following reaction? b. Would you expect epoxides to undergo similar reactions?arrow_forwardThe elimination reaction of butan-2-ol will produce butene and but-2-ene via mechanism a. SN1 O b. E2 O c. SN2 O d. E1arrow_forwardWhich of the provided schemes can be used to synthesize p-chlorophenol p-chlorophenol from benzen e? A. None of the below schemes are correct B. O C. D. SO3 H₂SO4 Cl₂ FeCl 903 H₂SO4 Product Product Product 1. NaOH 2. H₂O* SO 3 H₂SO4 Cl₂ FeCl Product Product Product Cl₂ FeCl H₂/Pd 1. NaOH 2. H₂O* p-Chlorophenol p-Chlorophenol p-Chlorophenolarrow_forward
- 3. For each of the reactions below, redraw the alkyl halide with the correct stereochemistry as indicated and provide mechanisms with explanations to account for the products observed. OCH, a. CH,OH R-enantiomer Racemic mixture b. NaCN H3C-Ç-CH2CH3 H3C-C-CH2CH3 Acetone CN Br R-enantiomer S-enantiomcr HIC HICarrow_forwardBromo compound N can undergo substitution with nucleophiles X to give mixtures of products O and P as shown below. Explain how this is possible, and suggest an explanation for the observation that the proportion of product P increases with increasing solvent polarity. F N Br X- O X ...X F Parrow_forward7. Give the stereochemistry for the product of each step of the conversion of trans-4-t- butylcyclohexanol to its cis isomer by the following reaction sequence. a. Reaction of the trans isomer with p-toluene sulfonylchloride in pyridine to compound A (C17H2603S). b. Reaction of A with sodium benzoate in dimethylformamidewith heat to compound B (C17H1402). c. Reaction of B with an aqueous solution of sodium hydroxide to give the cis isomer of the original starting material.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Coenzymes and cofactors; Author: CH15 SWAYAM Prabha IIT Madras;https://www.youtube.com/watch?v=bubY2Nm7hVM;License: Standard YouTube License, CC-BY
Aromaticity and Huckel's Rule; Author: Professor Dave Explains;https://www.youtube.com/watch?v=7-BguH4_WBQ;License: Standard Youtube License