
(a)
Interpretation:Thesynthesis of 1-chloropropane from propane as organic starting material needs to be explained with the use of other reagents.
Concept Introduction:
These functional groups involve in different
Substitution reaction of
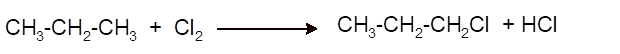
(b)
Interpretation: The synthesis of 2-chloropropane from propane as organic starting material needs to be explained with the use of other reagents.
Concept Introduction:
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester, or amide.
These functional groups involve in different chemical reactions such as substitution, addition, elimination etc.
Substitution reaction of alkanes are mainly used to form alkyl halide with the help of halogens.
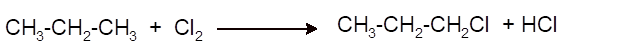
(c)
Interpretation: The synthesis of 1-bromopropane from propane as organic starting material needs to be explained with the use of other reagents.
Concept Introduction:
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester, or amide.
These functional groups involve in different chemical reactions such as substitution, addition, elimination etc.
Substitution reaction of alkanes are mainly used to form alkyl halide with the help of halogens.

(d)
Interpretation: The synthesis of 2-bromopropane from propane as organic starting material needs to be explained with the use of other reagents.
Concept Introduction:
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester, or amide.
These functional groups involve in different chemical reactions such as substitution, addition, elimination etc.
Substitution reaction of alkanes are mainly used to form alkyl halide with the help of halogens.

(e)
Interpretation: The synthesis of 1-iodopropane from propane as organic starting material needs to be explained with the use of other reagents.
Concept Introduction:
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester, or amide.
These functional groups involve in different chemical reactions such as substitution, addition, elimination etc.
Substitution reaction of alkanes are mainly used to form alkyl halide with the help of halogens.
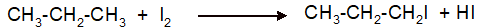
(f)
Interpretation: The synthesis of 2-iodopropane from propane as organic starting material needs to be explained with the use of other reagents.
Concept Introduction:
Functional groups are the groups of atoms or atoms which are bonded with parent carbon chain in the organic molecule and are responsible for the physical and chemical properties of the compound. In organic chemistry, there are different functional groups such as carboxylic acid, alcohol, ester, or amide.
These functional groups involve in different chemical reactions such as substitution, addition, elimination etc.
Substitution reaction of alkanes are mainly used to form alkyl halide with the help of halogens.
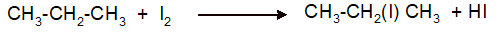
Trending nowThis is a popular solution!

Chapter 6 Solutions
Organic Chemistry: Structure and Function
- The following reaction conditions gives a mixture of two addition products A and B shown below. CI. HCI Ph Ph Ph Ph Ph A B via rearrangement via rearrangement >Draw< a clear mechanism using electron-flow arrows showing the formation of both products (A and B) from the starting materials. Transition states are not required and ignore the stereochemistry in this question. Do not use any other compounds in your answer except those given. Note that this is not a synthesis question. OYork rk.Unesk 020arrow_forward3. General Knowledge Rank the following compounds using numbers 1-4 in the box provided. (1 least) a. SN2 reactivity with NaOMe Br Br NH₂ b. SN1 reactivity with HBr HO OH c. E2 reactivity with KO'Bu Br Br Br Br d. Consider the following secondary halides. Changing the leaving group from fluoride to bromide would increase the rate of which of the following mechanisms? Circle all that apply. Br SN1 SN2 E1 E2 OH OH = most, 4 =arrow_forwardNeed help on this question help with parts a-b. Predict the major products.arrow_forward
- [Review Topics] [References] Devise a synthesis of butane using one of the starting materials and any of the reagents below using the fewest steps possible. If you need fewer than the 3 steps allowed, enter "none" for reagents in the remaining unused steps. Starting materials HCECH HOEC-CH, 1 Reagents 2 a Nanhbinh(D) b NaOH/H₂O c iodomethane Starting material Reagent for step 1 Reagent for step 2 Reagent for step 3 Submit Answer HCEC-CH₂CH₂ 3 diodoethane e 1-bromopropane. f 2-bromopropane Retry Entire Group HCEC-CH₂CH₂CH₂CH₂ 4 g 1-bromo-3-methylbutane h t-butyl bromide i H₂/Pd on carbon CH₂ HCEC-C-CH₂ H 5 9 more group attempts remaining KHINH (1) CH₂ HC=C-C-CH₂ CH₂ jH₂ Lindlar catalyst I Na/NH₂ (D 6 Previous Email Instructor Next Save and Exitarrow_forwardWhat is the Iupac name answers of all those questions from (d) till (h)???arrow_forward[Review Topics] [References] Draw the major organic product of the reaction shown below. Х OH + NaNH2 • You do not have to consider stereochemistry. Include counter-ions, e.g., Na+, I, in your submission, but draw them in their own separate sketcher. Separate structures with + signs from the drop-down menu. 9985 + [] ? ChemDoodle ?arrow_forward
- TROVIOW Topicaj (References) Draw the major organic product of the reaction shown below. HO, NANH2 You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Include cationic counter-ions, e.g., Na" in your answer, but draw them in their own sketcher. (Review Top Draw the major organic product of the reaction shown below. OH K2Cr207 H2SO4, H20 • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one.arrow_forwardPredict the major organic product of the given reaction. Select Draw Rings More 1. NaOEt/ETOH 2. CH3CH2CH2Br 3. dilute NaOH/heat 4. H3O* 5. Нeat d orrow mechanism for the scheme by adding any missing atoms, bonds, charges,arrow_forwardProvide the missing reagent to complete the following reaction. CH,CH,CH,ÔNa ? Write your answer on separate paper and submit a picture of your work here. Attach File Browse Local Files A Moving to another question will save this response.arrow_forward
- Are the statements (I-III) true descriptions of the reaction shown? Use the dropdown menu to give your answers. BH3, THF I The reaction does not involve rearrangement IL The reaction is stereospecific III The boron atom acts as a nucleophile I: choose your answer... II: choose your answer... II: choose your answer...arrow_forward3. Provide reagents/conditions to accomplish the following syntheses. More than one step is required in each case, and some reactions could lead to additional products other than that desired. Such an outcome is fine, because they can be separated based on their physical properties. The best answers for parts a, b, and c require ~3 operations, while part d requires -5 steps; answers with more than this number of steps that are correct will receive full credit. To assist with partial credit, drawing the structures of intermediate products would be beneficial. а) NH2 Br b) CI NO2 Но.arrow_forwardT hyperconjugation) to solve synthetic problems. (a) Write a detailed, electron-pushing mechanism that accounts for the formation of the organic El products. S Ⓒ N CH3 CI CH3 CH3 D CI NaOH E1 (b) A lesser-known elimination mechanism is represented here. In this mechanism, the base/nucleophile performs PT in the first step, but the leaving group does not immediately leave, as evidenced by the intermediate. Write a detailed, electron-pushing mechanism to describe this pathway. How does only one (1) organic product forming here compare to the two (2) products formed by El? CH3 NaOH PT CH3 + CH3 CH3 HOH CI CH3 CH3 + HOH O :CI:arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





