
Concept explainers
Interpretation: The important resonances of below molecules should be drawn.
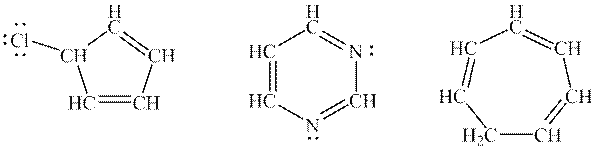
Concept introduction: When one single structure is unable to describe all the properties of single molecule, a phenomenon called resonance comes into play. This arises when two or more than two Lewis structures are possible for one molecule. All such structures are called resonating structures and have same placement of atoms in them but these have different locations of bond pairs and lone pairs. The resonating structures are inter-convertible with each other. The resultant of all the resonating or contributing structures is called the resonance hybrid.
Rules to form resonance structure are as follows:
1. Use arrow types 1 and 2 for resonance structure of anions in movement of negative charge.
2. Use only arrow type 3 to move a positive charge for resonance structure of cations.
3. The sigma bond should not be broken. Any atom must not move from its place and total number of electrons must be same in all resonance structures.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic Chemistry: A Guided Inquiry
- It is asking for second resonance structure. I think I did the second resonance structure wrong for each one. This is for organic chemistry.arrow_forwardCompare columns I and II, and write A if the value in column l is greater than in l B if the value in column 1l is greater than in ! cif both values are equal D if the value in column 1 cannot be compared with that in II Number of Resonance Structures Column II Column Iarrow_forwardIn the following Lewis structure of [BrO3]", every atom, bond and lone pair is positioned. To complete the structure, drag the formal charge tags to the appropriate atom(s). Each marker may be used more than once, or not at all. If an atom has a formal charge of zero, do not drag a tag to it. When you drag the marker in, place the little crosshairs in the upper left corner of the marker directly over the atom(s) in question (not above them). Ö: Br: 2+ 2-arrow_forward
- a) Draw all reasonable resonance structures for the following molecules. b) Label the major and minor resonance structures. If they are all equal write equal.arrow_forwardThe C=O double bond is called a “carbonyl bond.” Acetone and othercarbonyl compounds are introduced in some texts along with this structure a. Is this an important resonance structure of acetone? Explain. b. Does this structure convey any useful information about acetone? If so, what?arrow_forward3. Refer to the drawings below for this question about resonance. vul Compound A a. Draw all relevant resonance structures (i.e. all of the resonance structures that will contribute to the overall "look" of the molecule) for both Compound A and Compound B. conua וה Compound Barrow_forward
- Could you draw all valid resonance structrues for these two? I understand the rules for drawing resonance structures but I miss a lot of them.arrow_forwarddraw all the significant resonance structures of this moleculearrow_forwardDraw all possible resonance structures for the species belowarrow_forward
- 3. The following shows all resonance structures for the following molecule. a. Draw in all implied lone pairs. b. Draw in curved arrows that show the flow of electrons, making sure the arrows show the precise starting point and destination of the electrons. Label each arrow as: lp →→→л (p=lone pair) c. d. Rank the resonance structures from most stable to least based on the number of formal charges and atoms that lack an octet of electrons. ol-of-o. B A D d-d-d-o E C F Garrow_forwardDraw a curved arrow starting from the t bond of Draw all atoms of the resulting resonance structure C. Show resonance structure B that will form resonance charges and lone pairs. structure C. Select Draw Rings More Erase Select Draw Rings More Erase C C H :0 : I. H Q Q Resonance structure C is a major (more important) resonance structure. minor (less important) resonance structure.arrow_forwardCould you draw all valid resonance structures for this?arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
