
Chemistry: The Central Science (13th Edition)
13th Edition
ISBN: 9780321910417
Author: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4.6, Problem 4.16.2PE
The first stage of treatment at the reverse osmosis plant in Carlsbad, California, is to flow the water through rock, sand, and gravel as shown here. Would this step remove particulate matter? Would this step remove dissolved salts? [Section 18.4]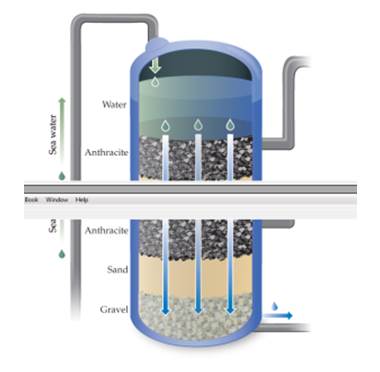
Expert Solution & Answer
Learn your wayIncludes step-by-step video

schedule02:10
Students have asked these similar questions
What mass of carbon dioxide is formed from burning one gallon of octane, C8H18? Octane has a density of 703 kg/m3.1 L = 0.2642 gal 1 lb = 453.592 gAn average tree absorbs 48 pounds of CO2 in one year. How many trees would you need to plant to offset the CO2produced from burning one gallon of octane every day for one year (365 days)?
What are Managerial Factors that could lead to failure of water treatment plant?
NEED HELP!
When potassium metal is placed in water, a large amount of energy is released as potassium hydroxide and hydrogen gas are produced in the reaction 2K(s) + 2H2O(l) → 2KOH(aq) + H2(g). Your lab partner says this is a redox reaction and a combustion reaction. Do you agree? Defend your answer by explaining whether or not it meets the requirements of each type of reaction.
Chapter 4 Solutions
Chemistry: The Central Science (13th Edition)
Ch. 4.1 - Prob. 4.1.1PECh. 4.1 - How are the boundaries between the regions of the...Ch. 4.2 - Air pollution in the Mexico City metropolitan area...Ch. 4.2 - Prob. 4.2.2PECh. 4.2 - Prob. 4.3.1PECh. 4.2 - Prob. 4.3.2PECh. 4.2 - Prob. 4.4.1PECh. 4.2 - Prob. 4.4.2PECh. 4.3 - Distinguish between photodissociation and...Ch. 4.3 - Prob. 4.5.2PE
Ch. 4.3 - Prob. 4.6.1PECh. 4.3 - Prob. 4.6.2PECh. 4.3 - Do the reactions involved in ozone depletion...Ch. 4.3 - Prob. 4.7.2PECh. 4.4 - Prob. 4.8.1PECh. 4.4 - Prob. 4.8.2PECh. 4.4 - Prob. 4.9.1PECh. 4.4 - Prob. 4.9.2PECh. 4.4 - Prob. 4.10.1PECh. 4.4 - Prob. 4.10.2PECh. 4.5 - Practice Exercise 2 The bond energy in N2 is 941...Ch. 4.5 - Prob. 4.11.2PECh. 4.5 - Prob. 4.12.1PECh. 4.5 - Prob. 4.12.2PECh. 4.5 - Prob. 4.13.1PECh. 4.5 - Prob. 4.13.2PECh. 4.5 - The figure shows the three lowest regions of...Ch. 4.5 - Prob. 4.14.2PECh. 4.6 - Where does the energy come from to evaporate the...Ch. 4.6 - Prob. 4.15.2PECh. 4.6 - Prob. 4.16.1PECh. 4.6 - The first stage of treatment at the reverse...Ch. 4.6 - Prob. 4.17.1PECh. 4.6 - Prob. 4.17.2PECh. 4 - Prob. 1DECh. 4 - Prob. 1ECh. 4 - Prob. 2ECh. 4 - Prob. 3ECh. 4 - Prob. 4ECh. 4 - Prob. 5ECh. 4 - Prob. 6ECh. 4 - Which of the following ions will always be a...Ch. 4 - Prob. 8ECh. 4 - Prob. 9ECh. 4 - Prob. 10ECh. 4 - Prob. 11ECh. 4 - List the common products formed when an organic...Ch. 4 - Prob. 13ECh. 4 - Prob. 14ECh. 4 - Prob. 15ECh. 4 - Prob. 16ECh. 4 - Prob. 17ECh. 4 - Prob. 18ECh. 4 - Prob. 19ECh. 4 - Prob. 20ECh. 4 - Prob. 21ECh. 4 - Prob. 22ECh. 4 - Prob. 23ECh. 4 - Prob. 24ECh. 4 - Prob. 25ECh. 4 - Prob. 26ECh. 4 - Prob. 27ECh. 4 - Prob. 28ECh. 4 - Prob. 29ECh. 4 - Explain, using Le Châtelier’s principle, why the...Ch. 4 - Prob. 31ECh. 4 - Prob. 32ECh. 4 - Prob. 33ECh. 4 - Prob. 34ECh. 4 - Prob. 35ECh. 4 - Prob. 36ECh. 4 - Prob. 37ECh. 4 - Prob. 38ECh. 4 - Prob. 39ECh. 4 - Prob. 40ECh. 4 - Prob. 41ECh. 4 - Prob. 42ECh. 4 - Prob. 43ECh. 4 - Prob. 44ECh. 4 - Prob. 45ECh. 4 - 18.85 The main reason that distillation is a...Ch. 4 - Prob. 47ECh. 4 - Prob. 48ECh. 4 - Prob. 49ECh. 4 - Prob. 50ECh. 4 - Prob. 51ECh. 4 - Prob. 52ECh. 4 - The process of iron being oxidized to make iron...Ch. 4 - At 1 atm pressure, CO2(s) sublimes at 78oC. Is...Ch. 4 - Prob. 55ECh. 4 - Prob. 56ECh. 4 - Prob. 57ECh. 4 - Prob. 58ECh. 4 - Prob. 59ECh. 4 - Prob. 60ECh. 4 - Using the standard molar entropies in Appendix C,...Ch. 4 - Which of these statements is true? All spontaneous...Ch. 4 - Prob. 63ECh. 4 - Prob. 64ECh. 4 - Prob. 65ECh. 4 - Prob. 66ECh. 4 - Prob. 67ECh. 4 - What is the temperature above which the Haber...Ch. 4 - Prob. 69ECh. 4 - Prob. 70ECh. 4 - Prob. 71ECh. 4 - Prob. 72ECh. 4 - Prob. 73ECh. 4 - Prob. 74ECh. 4 - Prob. 75ECh. 4 - Prob. 76ECh. 4 - As shown here, one type of computer keyboard...Ch. 4 - 19.3
a. What are the signs of ΔS and ΔH for the...Ch. 4 - Predict the signs of H and S for this reaction....Ch. 4 - Prob. 80ECh. 4 - The accompanying diagram shows how H (red line)...Ch. 4 - Prob. 82ECh. 4 - Prob. 83ECh. 4 - Prob. 84ECh. 4 - Prob. 85ECh. 4 - Prob. 86ECh. 4 - Prob. 87ECh. 4 - Can endothermic chemical reaction be spontaneous?...Ch. 4 - Prob. 89ECh. 4 - Prob. 90ECh. 4 - Prob. 91AECh. 4 - Prob. 92AECh. 4 - Prob. 93AECh. 4 - Prob. 94AECh. 4 - Prob. 95AECh. 4 - Prob. 96AECh. 4 - Prob. 97AECh. 4 - Prob. 98AECh. 4 - Prob. 99AECh. 4 - Prob. 100AECh. 4 - Prob. 101AECh. 4 - Prob. 102AECh. 4 - Prob. 103AECh. 4 - Alcohol-based fuels for automobiles lead to the...Ch. 4 - Prob. 105IECh. 4 - Prob. 106IECh. 4 - Prob. 107IECh. 4 - Prob. 108IECh. 4 - Prob. 109IECh. 4 - Prob. 110IECh. 4 - Prob. 111IECh. 4 - Prob. 112IECh. 4 - Although there are many ions in seawater, the...Ch. 4 - The Ogallala aquifer described in the Close Look...Ch. 4 - Prob. 115IE
Additional Science Textbook Solutions
Find more solutions based on key concepts
A compound that contains only C and H was burned in excess O2 to give CO2 and H2O. When 0.270 g of the compound...
General Chemistry: Atoms First
Nitric acid in acid rain forms when gaseous nitrogen dioxide pollutant reacts with gaseous oxygen and liquid wa...
Introductory Chemistry (6th Edition)
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
Draw the structures of straight-chain alcohols that have from one to six carbons with an OH group at the end of...
Essential Organic Chemistry (3rd Edition)
The content of is 15.5g/100 mLblood. The molar mass of hemoglobin is about 64,500 g/mol, and there are four ion...
General Chemistry: Principles and Modern Applications (11th Edition)
10.1 Indicate whether each of the following statements is characteristic of an acid, a base, or
both:
has a so...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Cyanobacteria are chemosynthetic organisms that reside deep in the ocean near hydrothermal vents. Archeoglobus, one of these types of cyanobacteria, uses the pathway shown in the chemical equation. 4H2 + SO42− + 2H+ → H2S + 4H2O Based on this equation, which explanation best describes how the process of chemosynthesis in bacteria differs from photosynthesis, and why can this process occur in the absence of oxygen? Chemosynthesis uses H2 as an energy source, while photosynthesis uses the Sun as an energy source. Chemosynthesis uses H+ as an energy source, while photosynthesis uses the Sun as an energy source. Chemosynthesis reduces a sulfate molecule in the absence of oxygen and light, while photosynthesis depends on light to reduce carbon dioxide. Chemosynthesis oxidizes a sulfate molecule in the absence of oxygen and light, while photosynthesis depends on light to reduce carbon dioxide.arrow_forwardWhat are the practices that are considered waste minimization and pollution prevention?arrow_forwardDiiodine pentaoxide oxidizes carbon monoxide to carbon dioxide under room conditions, yielding iodine as the sec- ond product: 1,O3(s) + 5 CO(g) → L(s) + 5 CO2(g) This can be used in an analytical method to measure the amount of carbon monoxide in a sample of air. Determine the oxidation numbers of the atoms in this equation. Which species is oxidized and which is reduced?arrow_forward
- In Module #4, we discussed the role of chlorofluorocarbons (CFCs) in catalyzing the destruction of O3 in the stratosphere. One example of a CFC that is known to exasperate formation of the hole in the O3 layer is dichlorodifluoromethane (CF2Cl2). In the year 2000, this chemical reached its peak concentration of 2.2 x 10-11 M in the atmosphere. The destruction of CF2Cl2 can be approximated as a first-order process and the 1996 treaty on CFC emissions has eliminated its release into the atmosphere. Given that the rate constant for destruction of CF2Cl2 in the atmosphere is 0.009 yr-1, how long will it take for the concentration of CF2Cl2 to decline to 0.22 x 10-11 M?arrow_forwardThe research group has recently focused on identifying the chemical properties of fluorocarbons, such as CF4 and C2F4. These fluorocarbons display a very different reactivity compared to hydrocarbons. Teflon (C2F4)n is a fluoropolymer. When Teflon is mixed with silica at high temperature, all solid decomposes into gases. Write down the reactions that can take place during the process and explain your reasoning.arrow_forwardReactants: Sodium Hydroxide (NaOH) Potassium Permanganate (KMnO4) Glucose (C₆H₁₂O₆) What is the purpose of the sodium hydroxide in the redox reaction experiment?arrow_forward
- Ff.203. what is meant by a hydrogenated oil? Why is this potentially dangerous to human health?arrow_forward19. The chemical equation for photosynthesis is _CO2 (g) + _H₂O (1)→ __C6H1206 (s) + (C6H12O6 is glucose, the simplest sugar) a. Balance the chemical equation above by filling in the blanks. b. According to onetreeplanted.org, the average tree (across all climates and sizes) removes 10 kg of carbon dioxide from the atmosphere every year. Based on this, what mass of glucose is produced annually by the average tree? O2 (g) c. Teak is a common species of tropical tree. The graph below shows the amount of CO₂ absorbed by teak forests in tons of CO₂ per hectare of forest per year, according to the climate region where the forests are found (dry = low rainfall, humid=high rainfall). Looking at your photosynthesis equation again, what does this data suggest about which reactant is the limiting reactant for teak trees? Explain. C Teak Carbon sequestration rate (tons CO₂ ha -1yr -1) 10 10 20 Tropical humid Tropical dry Aboveground biomass, 0-20 years Belowground biomass, 0 -20 yearsarrow_forwardBalance the chemical equation. Au + HCI + HNO3→AUCI3 + NO + H20 Assume the coefficient of H20 is 2. What is the balanced equation? DAu +HCI +HNO3-[Auc13 +NO +H20arrow_forward
- Write your complete solutions on yellow paper, round your answers to the appropriate number of significant figures, and box the final answer. The deadline for submission is at the end of the class. Note: You do not need to write your solutions for the items that can be derived from your calculators. 1. Three students (Saerom, Hayoung, and Jiwon) are asked to determine the ppm Ca in a drinking water sample by titration with EDTA. Each student performed five trials for the sample. The results in ppm Ca are the following: Saerom Hayoung Jiwon a. Get the mean, SD, and % RSD of each student's results. b. If the true value is 35 ppm, calculate the relative error of each student's results. c. Comment on the precision and accuracy of each student's results. The maximum % RSD and the relative error set by the lab is 5.0%. d. Use the following table to summarize your answers. Mean SD %RSD Relative Error Comment 31, 32, 33, 33, 32 35, 34, 34, 35, 34 35, 38, 33, 35, 37 Saerom Mean SD 95% CI…arrow_forwardFlint water crisis: “Soon after the city began supplying residents with Flint River water in April 2014, residents started complaining that the water from their taps looked, smelled, and tasted foul. Despite protests by residents lugging jugs of discoloured water, officials maintained that the water was safe. A study conducted the following year by researchers at Virginia Tech revealed the problem: Water samples collected from 252 homes through a resident-organized effort indicated citywide lead levels had spiked, with nearly 17% of samples registering above the federal “action level”. More than 40% measured above 5 ppb of lead, which the researchers considered an indication of a “very serious” problem.” Lead (Pb) was released from the distribution pipeline in the city of Flint due to a switch of the water source. (1) Please use Gibb’s formation energy in the table to find out the equilibrium constants for each reaction (R1-R4). (2) What will be total dissolved lead at pH 8 in pure…arrow_forwardIf you have a gas furnace it is important to make sure that it is tuned up with a proper flow of oxygen. Without a proper flow of oxygen to the furnace, it can burn the natural gas (CH4) with some incomplete combustion. Why can it be so dangerous if your furnace is burning natural gas with incomplete combustion?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning
ENVIRONMENTAL POLLUTION; Author: 7activestudio;https://www.youtube.com/watch?v=oxtMFmDTv3Q;License: Standard YouTube License, CC-BY