
Concept explainers
Read Appendix B on naming bicyclic compounds. Then give IUPAC name for each of the following compounds.
a. 
b. 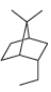
c. 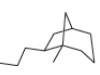
d. 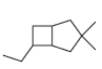
(a)
Interpretation: The IUPAC name for the given bicyclic compound is to be stated.
Concept introduction: The root name of bicyclic compounds depends upon the number of carbon atoms present in the rings. The naming of bicyclic compounds is done as,
(1) The total number of carbon atoms in the molecule is to be counted. It represents the root name.
(2) The number of carbon atoms between the bridgeheads is to be counted and placed in square brackets in descending order.
(3) The word bicyclo is placed at the beginning of the name.
The general name such compounds is represented as
Answer to Problem 4.75P
The IUPAC name for the given bicyclic compound is
Explanation of Solution
The given bicyclic compound is shown below.
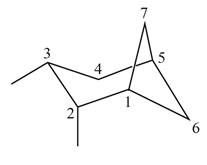
Figure 1
The given compound contains seven carbon atoms in the ring which are single bonded to each other. Therefore, the root name is heptane. In this case, there are
The IUPAC name for the given bicyclic compound is
(b)
Interpretation: The IUPAC name for the given bicyclic compound is to be stated.
Concept introduction: The root name of bicyclic compounds depends upon the number of carbon atoms present in the rings. The naming of bicyclic compounds is done as,
(1) The total number of carbon atoms in the molecule is to be counted. It represents the root name.
(2) The number of carbon atoms between the bridgeheads is to be counted and placed in square brackets in descending order.
(3) The word bicyclo is placed at the beginning of the name.
The general name such compounds is represented as
Answer to Problem 4.75P
The IUPAC name for the given bicyclic compound is
Explanation of Solution
The given bicyclic compound is shown below.
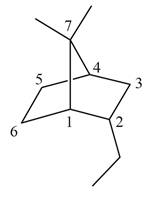
Figure 2
The given compound contains seven carbon atoms in the ring which are single bonded to each other. Therefore, the root name is heptane. In this case, there are
The IUPAC name for the given bicyclic compound is
(c)
Interpretation: The IUPAC name for the given bicyclic compound is to be stated.
Concept introduction: The root name of bicyclic compounds depends upon the number of carbon atoms present in the rings. The naming of bicyclic compounds is done as,
(1) The total number of carbon atoms in the molecule is to be counted. It represents the root name.
(2) The number of carbon atoms between the bridgeheads is to be counted and placed in square brackets in descending order.
(3) The word bicyclo is placed at the beginning of the name.
The general name such compounds is represented as
Answer to Problem 4.75P
The IUPAC name for the given bicyclic compound is
Explanation of Solution
The given bicyclic compound is shown below.
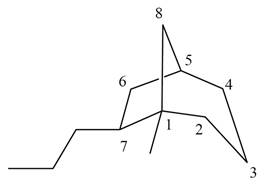
Figure 3
The given compound contains eight carbon atoms in the ring which are single bonded to each other. Therefore, the root name is octane. In this case, there are
The IUPAC name for the given bicyclic compound is
(d)
Interpretation: The IUPAC name for the given bicyclic compound is to be stated.
Concept introduction: The root name of bicyclic compounds depends upon the number of carbon atoms present in the rings. The naming of bicyclic compounds is done as,
(1) The total number of carbon atoms in the molecule is to be counted. It represents the root name.
(2) The number of carbon atoms between the bridgeheads is to be counted and placed in square brackets in descending order.
(3) The word bicyclo is placed at the beginning of the name.
The general name such compounds is represented as
Answer to Problem 4.75P
The IUPAC name for the given bicyclic compound is
Explanation of Solution
The given bicyclic compound is shown below.
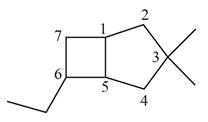
Figure 4
The given compound contains seven carbon atoms in the ring which are single bonded to each other. Therefore, the root name is heptane. In this case, there are
The IUPAC name for the given bicyclic compound is
Want to see more full solutions like this?
Chapter 4 Solutions
Organic Chemistry
Additional Science Textbook Solutions
General Chemistry: Principles and Modern Applications (11th Edition)
Fundamentals of Heat and Mass Transfer
General, Organic, and Biological Chemistry - 4th edition
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
- 1. Provide the IUPAC name for each of the following organic molecules. a. b. C. H3C H3C, Home H3C CH3 CH3 CH3 Br H 2. Draw structures for the following organic molecules, using the wedged/hashed line convention to depict any stereochemical relationships between groups: a. cis-3-ethylcyclobutane-1-carbaldehyde b. (R)-1-chloro-6-iodo-6-methyloctan-3-onearrow_forwardWhich heteroatom don't contain heterocyclic compounds? a. Phosphorus b. Oxygen c. Nitrogen d. Sulfurarrow_forwardRank the following compounds in order from smallest heat of combustion to largest heat of combustion. A Barrow_forward
- Which compound is second least stable, giving off the second most heat of hydrogenation or combustion (when burned)? a. O b. d. Of.arrow_forwardProvide the IUPAC name for the following. You may use either the 'prefix-2-alkene' or the 'prefixalk-2-ene' naming convention, but don't forget to include any stereoisomerism in your name if relevant. a. C. Br CH₂ b. farrow_forward1. Give the IUPAC name for the provided structures a. thy equ b. d. CIarrow_forward
- 1. What is the relationship between the following two compounds?a.identical b.not isomers; different compounds entirely c.constitutional isomers2. Is the following alkene cis, trans, or neither?arrow_forwardHydrocarbons like benzene are metabolized in the body to arene oxides, which rearrange to form phenols. This is an example of a general process in the body, in which an unwanted compound (benzene) is converted to a more water-soluble derivative called a metabolite, so that it can be excreted more readily from the body. a.) Classify each of these reactions as oxidation, reduction, or neither.b.) Explain why phenol is more water soluble than benzene. This means that phenol dissolves in urine, which is largely water, to a greater extent than benzenearrow_forwardGive the correct IUPAC name and structure of the ff.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

