
Concept explainers
Which compound in each pair has the higher boiling point?
a. 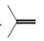 or
or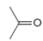 c.
c. 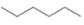 or
or
b. 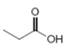 or
or 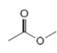 d.
d. 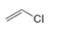 or
or 
(a)
Interpretation: In the given pair, the compound with the higher boiling point is to be predicted.
Concept introduction: Boiling point is defined as the temperature at which the compound is converted to a gaseous state.
The boiling point of compounds depends upon the molecular weight and the intermolecular forces present in the compounds. The intermolecular forces are the forces that exist in between the two or more atoms in a compound. Due to strong intermolecular forces, the molecule will require more energy to break the bonds. The compounds that have ionic bonding and hydrogen bonding will possess higher boiling point.
Answer to Problem 3.13P
In the given pair of compounds,
Explanation of Solution
The given compounds are
In
The increasing order of boiling point with intermolecular forces is,
Hence, the boiling point of
In the given pair of compounds,
(b)
Interpretation: In the given pair, the compound with the higher boiling point is to be predicted.
Concept introduction: Boiling point is defined as the temperature at which the compound is converted to a gaseous state.
The boiling point of compounds depends upon the molecular weight and the intermolecular forces present in the compounds. The intermolecular forces are the forces that exist in between the two or more atoms in a compound. Due to strong intermolecular forces, the molecule will require more energy to break the bonds. The compounds that have ionic bonding and hydrogen bonding will possess higher boiling point.
Answer to Problem 3.13P
In the given pair of compounds,
Explanation of Solution
The given compounds are
In
In the given pair of compounds,
(c)
Interpretation: In the given pair, the compound with the higher boiling point is to be predicted.
Concept introduction: Boiling point is defined as the temperature at which the compound is converted to a gaseous state.
The boiling point of compounds depends upon the molecular weight and the intermolecular forces present in the compounds. The intermolecular forces are the forces that exist in between the two or more atoms in a compound. Due to strong intermolecular forces, the molecule will require more energy to break the bonds. The compounds that have ionic bonding and hydrogen bonding will possess higher boiling point.
Answer to Problem 3.13P
In the given pair of compounds,
Explanation of Solution
The given compounds are
In both the compounds,
In the given pair of compounds,
(d)
Interpretation: In the given pair, the compound with the higher boiling point is to be predicted.
Concept introduction: Boiling point is defined as the temperature at which the compound is converted to a gaseous state.
The boiling point of compounds depends upon the molecular weight and the intermolecular forces present in the compounds. The intermolecular forces are the forces that exist in between the two or more atoms in a compound. Due to strong intermolecular forces, the molecule will require more energy to break the bonds. The compounds that have ionic bonding and hydrogen bonding will possess higher boiling point.
Answer to Problem 3.13P
In the given pair of compounds,
Explanation of Solution
The given compounds are
Van der Waals forces exist in both the compounds due to the presence of
In the given pair of compounds,
Want to see more full solutions like this?
Chapter 3 Solutions
Organic Chemistry
- Draw a stepwise mechanism for the attached reaction that forms ether D. D can be converted to the antidepressant fluoxetine (trade name Prozac) in a single steparrow_forwardGive the IUPAC name for each sulde.arrow_forwardRank the compounds in attached each group in order of increasing boiling point ?arrow_forward
- Which compound is not aromatic? A. only A and D B. A, B and C C. only B D. only Aarrow_forward-.8 Identify the compound in each group that is most soluble in water. Explain. a. butanone, butanoic acid, butane b. ethanoic acid (acetic acid), hexanoic acid, octanoic acid ciation ofarrow_forwardWhich of the following are a. hemiacetals? b. acetals? c. hydrates?arrow_forward
- Draw the following: 1. a. 3-methyl-1-butanethiol (skunk scent) b. triphenylmethanol c. 4-(bromomethyl)-3-octanol d. 3-cyclopentenethiolarrow_forwardexplain the following observations a. propanol is more water soluble than propane b. propanol is more water soluble than propanonearrow_forward2. How many different ß-hydroxyaldehydes and ß-hydroxyketones, including constitutional isomers and stereoisomers, are formed upon treatment of a mixture of acetone and benzaldehyde with base? a. b. 2 c. 3 d. 4arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
