
Using the data of Figure 3.10, predict the sequence of electron carriers in the membrane of an organism growing aerobically that has the following electron carriers: ubiquinone, cytochrome aa3, cytochrome b, NADH, cytochrome c, FAD.
To predict:
The sequence of electron carriers such as ubiquinone, cytochrome aa3, cytochrome b, NADH, cytochrome c, and FAD in the membrane of an organism growing aerobically by using the data of Figure 3.10 in the text book.
Concept introduction:
An oxidation is a process in which electrons are removed from a substance whereas, in reduction process, electrons are added to the substance. The combination of these processes is termed as redox (oxidation-reduction) reaction.
Explanation of Solution
The electron transport chain consists of carriers of protons and electrons, which facilitate their transfer from a primary electron donor to a molecule that acts as the terminal acceptor.
The differences in the redox reaction can be identified by comparing the standard reduction potential (E0’). If E0’ of the substance is more negative than the other, it is identified that it undergoes reduction and if E0’ of the substance is more positive, that means it accepts electron and undergoes an oxidation process.
The sequence of electron carriers in the membrane of an organism growing aerobically and the following electron carriers produced as follows:
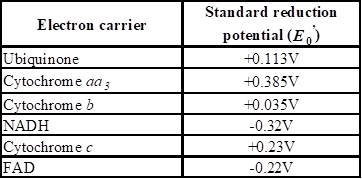
Want to see more full solutions like this?
Chapter 3 Solutions
Brock Biology of Microorganisms (15th Edition)
- Create a visual representation, or a diagram, of the concepts in respiration and photosynthesis. You are NOT simply creating a list of terms with definitions, but a diagram of the process. Use ALL of these terms in the diagram/representation. Acetyl CoA --- Aerobic --- Anaerobic --- Citrate --- Citric Acid Cycle --- CO2, H2O, O2, ATP, ADP, Pi, NADH, NAD+, FADH2, FAD, GTP, and GDP --- Coenzyme --- Cytoplasm --- Electron Transport Chain --- Ethanol --- Fermentation --- Glucose --- Glycolysis --- Inner Mitochondrial Membrane --- Lactic Acid --- Mitochondrial Matrix --- Oxaloacetate --- Pyruvate --- Pyruvate Oxidationarrow_forwardWhy are electron carriers (NAD+/NADH and FAD/FADH2) so important in the process of cellular respiration? A)NADH and FADH2 are major components of the ETC, so without them, there would be no ETC in the cell. B)They deliver electrons to the ETC, which in turn sets up chemiosmosis, where most of the ATP is generated. C)They separate the electrons from the protons so that the protons can be moved out of the mitochondrion. D)The electrons that they carry are able to directly phosphorylate ADP in order to generate the bulk of ATP in the cell. E) They transport protons across the mitochondrial membrane. 14.arrow_forwardWhich of the following sequence of proteins and the corresponding number of pumped protons is correct during the electron transport chain from NADH? Ubiquinone → Complex III: 4 H+ → Cytochrome c→ Complex IV: 2 Ubiquinone Complex III: 2 H+→ Cytochrome c→ Complex IV:2 Ubiquinone → Cytochrome c→ Complex III: 4 H+ → Complex IV: 2 D. Complex 1: 4 H+ → Ubiquinone → Complex III: 2H+ → Complex IV: 2 H+ → Cytochrome c A. Complex I: 4 H+ → B. Complex I: 4 H+ C. Complex I: 4 H+arrow_forward
- If the order for the electron transport chain starting from NADH to Oxygen is this: NADH-->Flavin mononucleotide (FMN)--> Coenzyme Q (CoQ) --> Cytochrome b (Cyt b)--> Cytochrome c (Cyt C)--> Cytochrome a3 (cyt a3) --> Oxygen (O2) Then, what is the order for the electron transport chain starting with FADH2 to oxygen (O2)?arrow_forward5. a) The cell creates molecules of NADH and FADH2to use in the electron transport chain as they are electron carrier molecules. The electron transfers from these moleculesdrives the movement of what molecule across the mitochondrial membrane?b) What is chemiosmosis?c) How does the cell use chemiosmosis to drive overcome the thermodynamic barrier of bringing phosphates close togehter to create ATP?d) What steps of oxidative phosphorylation are in the cytosol and which are in the mitochondria?e) Cellular respiration can be controlled at the post-translational level. What does this mean, using GLUT as an example? Why is post-translational control of GLUT evolutionarilyadaptive over transcriptional control?f) How does establishment of equillibria of various molecules control metabolic reactions? What are three ways high ATP in the cell control cellular respiration?g) Why are the enzymes controlling the initial steps of various biochemical pathways are targeted by allosteric control?arrow_forwardFor each statement below, write whether the event occurs in GLYCOLYSIS, KREBS CYCLE, or ELECTRON TRANSPORT CHAIN. 1. Oxygen is the final acceptor of electrons 2. Occurs outside the mitochondria 3. Produces FADH2 4. Begins with glucose 5. Occurs in the mitochondrial matrix 6. Ends with pyruvic acid 7. Where chemiosmosis occurs 8 .Forms water 9.Occurs in the inner membrane of the mitochondria 10. Produces the most ATParrow_forward
- The following statements describe the path of electrons from NADH through the electron transport chain. Please arrange them in order: Cytochrome c (Fe2+) carries electrons to Complex IV. Cytochrome c (Fe3+) is reduced to cytochrome c (Fe2+) QH2 carries electrons to Complex III Oxygen is reduced to water Ubiquinone (coenzyme Q) is reduced to ubiquinol (QH2)arrow_forwardTable 8: ATP Yield from Anaerobic Respiration (for 1 Glucose Molecule) # of ATPS Generated through Substrate-Level Phosphorylation # of ATPS Generated from NADH and FADH, through Oxidative # of NADH and FADH, Produced Gross Subtotal of ATPS Produced Process Phosphorylation NADH: FADH2: Glycolysis NADH: FADH2: GROSS TOTAL of ATPS Produced # of ATPS Spent NETTOTAL of ATPS Produced Fermentationarrow_forwardIndicate whether the statement is true for aerobic respiration, photosynthesis or both: NADH is oxidized to NAD+ at complex I a)Aerobic Respiration b)Photosynthesis c)Botharrow_forward
- Which of the following statements concerning the complete oxidation of FADH2 in the electron transport chain is NOT true? a. In the final step, electrons from cytochrome c to O2 reducing it to H2O in complex IV, and four protons are transported from the intermembrane space to the matrix. b. In the first step, electrons from FADH2 are transferred in complex II to ubiquinone, which does not transport any proton across the inner mitochondrion membrane. c. In the second step, complex III transfers the electrons from ubiquinone to cytochrome c, and four protons are transported from the matrix to the intermembrane space. d. The complete oxidation of FADH2 causes transfer of 6 protons and yields two ATP.arrow_forwardChoose the correct path taken by a pair of electrons as they travel down the electron-transport chain. OA) NADH → Complex CoQ → Complex III → Cyt c→ Complex IV → 02 OB) FADH2 → Complex I CoQ - Complex III → O c) Cyt c Complex IV → 02 - 1 NADH Complex I Complex II Complex III 02 D) FADH2 → Complex II CoQ Complex III → Cyt c → Complex IV → 02 OE) A and D. Cyt c Complex IV → -arrow_forwardExplain how would compounds such as NAD+, NADH, ubiquinone, ubiquinol and cytochrome are able to facilitate the transport of electrons in the electron transport chain. Provide an explanation for why this mechanism for transporting electrons is evolutionary conserved across species and across biological systems.arrow_forward
 Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON
Human Anatomy & Physiology (11th Edition)BiologyISBN:9780134580999Author:Elaine N. Marieb, Katja N. HoehnPublisher:PEARSON Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education,
Anatomy & PhysiologyBiologyISBN:9781259398629Author:McKinley, Michael P., O'loughlin, Valerie Dean, Bidle, Theresa StouterPublisher:Mcgraw Hill Education, Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company
Molecular Biology of the Cell (Sixth Edition)BiologyISBN:9780815344322Author:Bruce Alberts, Alexander D. Johnson, Julian Lewis, David Morgan, Martin Raff, Keith Roberts, Peter WalterPublisher:W. W. Norton & Company Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co.
Laboratory Manual For Human Anatomy & PhysiologyBiologyISBN:9781260159363Author:Martin, Terry R., Prentice-craver, CynthiaPublisher:McGraw-Hill Publishing Co. Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education
Inquiry Into Life (16th Edition)BiologyISBN:9781260231700Author:Sylvia S. Mader, Michael WindelspechtPublisher:McGraw Hill Education





