
Concept explainers
(a)
Interpretation:
In conjugated triene,
Concept introduction:
The molecular orbital is a combination of two atomic orbitals. It is used to represent the regions in a molecule where the electron is likely to be present in an orbital. It represents the wave-like nature of an electron in a molecule. It may be symmetric or antisymmetric. It may be bonding, antibonding or non-bonding. It may be HOMO or LUMO.
Answer to Problem 28.2P
In
Explanation of Solution
The structure of
The electron configuration of carbon atom is
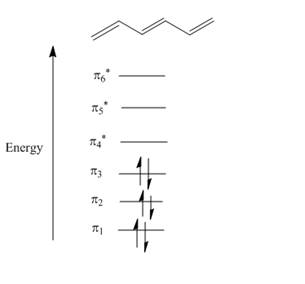
Figure 1
The atomic p orbital of carbon atom overlapped together to form molecular orbital. A total of six molecular orbitals are formed due to six carbon atomic orbital overlapping in
(b)
Interpretation:
Each molecular orbital is to be classified as symmetric and antisymmetric.
Concept introduction:
The molecular orbital is a combination of two atomic orbitals. It is used to represent the regions in a molecule where the electron is likely to be present in an orbital. It represents the wave-like nature of an electron in a molecule. It may be symmetric or antisymmetric. It may be bonding, antibonding or non-bonding. It may be HOMO or LUMO.
Answer to Problem 28.2P
The molecular orbitals
Explanation of Solution
In molecular orbital theory, the MO is said to be symmetric or anti-symmetric is depend on the relative phase of the two terminal carbons. In symmetric MO, the peaks reflect across the reference plane into the peaks and troughs reflect into troughs. On the other hand, in antisymmetric MO, the peaks reflect into troughs and vice versa. According to the general principle, the even number molecular orbitals are antisymmetric and odd number molecular orbitals are symmetric. Therefore, the molecular orbitals
The odd number of molecular orbitals is symmetric molecular orbitals that are
(c)
Interpretation:
Bonding and antibonding molecular orbitals are to be identified.
Concept introduction:
The molecular orbital is a combination of two atomic orbitals. It is used to represent the regions in a molecule where the electron is likely to be present in an orbital. It represents the wave-like nature of an electron in a molecule. It may be symmetric or antisymmetric. It may be bonding, antibonding or non-bonding. It may be HOMO or LUMO.
Answer to Problem 28.2P
The molecular orbital
Explanation of Solution
In
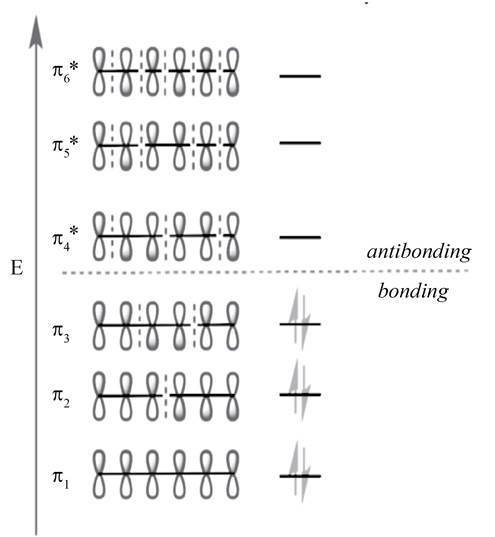
Figure 2
The molecular orbital with lower energy are bonding MO
(d)
Interpretation:
Among the molecular orbitals, the frontier molecular orbital is to be identified.
Concept introduction:
The molecular orbital is a combination of two atomic orbitals. It is used to represent the regions in a molecule where the electron is likely to be present in an orbital. It represents the wave-like nature of an electron in a molecule. It may be symmetric or antisymmetric. It may be bonding, antibonding or non-bonding. It may be HOMO or LUMO.
Answer to Problem 28.2P
The molecular orbital
Explanation of Solution
The highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) is known as frontier molecular orbitals. In
The molecular orbital
(e)
Interpretation:
Whether the phase at terminal carbons in HOMO orbitals is the same or different is to be stated.
Concept introduction:
The molecular orbital is a combination of two atomic orbitals. It is used to represent the regions in a molecule where the electron is likely to be present in an orbital. It represents the wave-like nature of an electron in a molecule. It may be symmetric or antisymmetric. It may be bonding, antibonding or non-bonding. It may be HOMO or LUMO.
Answer to Problem 28.2P
The HOMO orbital that is
Explanation of Solution
According to the general principle, the even number molecular orbitals are antisymmetric or its phase terminal carbons are different and odd number molecular orbitals are symmetric or its phase terminal carbons are same. The HOMO orbital is
The HOMO orbital is
(f)
Interpretation:
Whether the phase at terminal carbons in LUMO orbitals is the same or different is to be stated.
Concept introduction:
The molecular orbital is a combination of two atomic orbitals. It is used to represent the regions in a molecule where the electron is likely to be present in an orbital. It represents the wave-like nature of an electron in a molecule. It may be symmetric or antisymmetric. It may be bonding, antibonding or non-bonding. It may be HOMO or LUMO.
Answer to Problem 28.2P
The LUMO orbital that is
Explanation of Solution
According to the general principle, the even number molecular orbitals are antisymmetric or its phase terminal carbons are different and odd number molecular orbitals are symmetric or its phase terminal carbons are same. The LUMO orbital is
The LUMO orbital is
Want to see more full solutions like this?
Chapter 28 Solutions
Organic Chemistry
- (a) Draw the orbital picture for diazomethane showing the explicit overlap of the contributing AO's. (b) Draw the MO energy diagram for diazomethane and identify the HOMO and LUMO.arrow_forward18. Amines are like ammonia in being weak bases. They do this by using their unshared electron pair to accept a proton. (a.) Show the reaction that would take place between trimethyl amine and HCI. (b) What hybridization state would you expect for the nitrogen atom in the product of this reaction?arrow_forward(a) Using only the valence atomic orbitals of a hydrogenatom and a fluorine atom, and following the model ofFigure 9.46, how many MOs would you expect for the HFmolecule? (b) How many of the MOs from part (a) would beoccupied by electrons? (c) It turns out that the difference inenergies between the valence atomic orbitals of H and F aresufficiently different that we can neglect the interaction ofthe 1s orbital of hydrogen with the 2s orbital of fluorine.The 1s orbital of hydrogen will mix only with one 2p orbitalof fluorine. Draw pictures showing the proper orientationof all three 2p orbitals on F interacting with a 1s orbitalon H. Which of the 2p orbitals can actually make a bondwith a 1s orbital, assuming that the atoms lie on the z-axis?(d) In the most accepted picture of HF, all the other atomicorbitals on fluorine move over at the same energy into themolecular orbital energy-level diagram for HF. These arecalled “nonbonding orbitals.” Sketch the energy-level diagramfor HF using…arrow_forward
- Q. 3. (a) Determine the orbital in which the 9th and 10th electron will go in case of N2 molecule and its monopositive ion based on MO diagram. (b) Determine the pH of a 2 M ammonia solution, having the following balanced chemical reaction as follows (Kb=1.5×104) NH3(aq) + H2O(1) = NH,*(aq) + OH (aq) (c) Identify titration curves given below. Also explain the behavior and shape of the curve they followed during the following titration. A A Conductance Conductance В i B Volume of titrant Volume of titrantarrow_forwardAnswer the following questions for 1,3,5- hexatriene, the conjugated triene containing six carbons. Which MOs are the frontier molecular orbitals?arrow_forward(a)The concept of hybridization was first proposed by Linus Pauling in 1931, that explain about the concept of mixing orbitals to form new hybrid orbital suitable for the pairing of electrons to form chemical bonds. Draw the kekule structure of a compound that contains only carbon and hydrogen atoms that has: (1) One sp hybridize carbon, two sp? hybridized carbons and two sp hybridized carbon (ii) Two sp' hybridized carbon and two sp hybridized carbons (ii) Two sp? hybidized carbons and two sp hybydized carbonarrow_forward
- The unsaturation number or degree of unsaturation (U) can be used to determine the number of rings and multiple bonds in a compound from its molecular formula. Given a structure, you can determine the number of hydrogens without having to count them explicitly. Consider three compounds and their degree of unsaturation. (a) A compound A has the molecular formula C7H13ClN2OC7H13ClN2O. How many rings and/or π bonds does it contain?arrow_forwardTOPIC: QUANTUM, ATOMIC AND MOLECULAR PHYSICIS (a) Using Hückel's method, describe the energy of the molecular orbitals T of cyclobutadiene C4H4, which is a flat molecule made up of a square ring of carbons, as shown in the figure; (b) Do the same for 1,3-butadiene C4H6, which is an elongated planar molecule, as shown in the figure. (c) In both cases, draw an energy diagram indicating the filling of the orbitals and identifying the HOMO and the LUMO. Sketch by hand schematically (using two colors to distinguish the lobes from the 2pz orbitals) the result obtained for the orbitals, identifying them with the corresponding energy. If possible, you can use the Orbital Viewer program to plot the orbitals.arrow_forwardDraw orbital pictures of the pi bonding in the following compounds:(a) CH3COCH3 (b) HCN (c) CH2“CH¬CHCHCNarrow_forward
- Hybridization. (a) For acetone (shown below), give the electronic configuration as a function of energy and show how the orbitals are modified to explain sp² hybridization on the carbonyl oxygen. (b) Place the appropriate number of electrons into the hybridized orbitals to explain the bonding (geometry and types of orbitals) around the oxygen atom. (c) Describe the bonding to justify the sp² hybridization of the oxygen atom. I Carrow_forwardThe degree of unsaturation, or index of hydrogen deficiency, is the number of pi bonds plus rings in a molecule.Specify the degree of unsaturation (index of hydrogen deficiency) of the following formulas:(a) C9H12(b) C14H18(c) C10H8N2arrow_forwardHow many nodes are there in each of the MOs of 1,3-butadiene?arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

