
a)
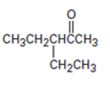
Interpretation:
The preparation of the compound shown using acetoacetic ester synthesis is to be given.
Concept introduction:
Acetoacetic ester synthesis converts an
To give:
The preparation of the compound shown using acetoacetic ester synthesis.
b)
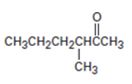
Interpretation:
The preparation of the compound shown using acetoacetic ester synthesis is to be given.
Concept introduction:
Acetoacetic ester synthesis converts an alkyl halide in to a methyl ketone having three more carbons. The methyl ketone part comes from acetoacetic eater while the remaining carbon comes from the primary alkyl halide. The reaction occurs in three steps i) enolate ion formation ii) SN2 attack of the enolate anion on the alkyl halide iii) hydrolysis and decarboxylation.
To give:
The preparation of the compound shown using acetoacetic ester synthesis.
Trending nowThis is a popular solution!

Chapter 22 Solutions
Organic Chemistry
- Propose a mechanism for the followingreaction → NaOH,acetonearrow_forwardWhat products are formed by acidic hydrolysis of the following compound? benzaldehyde and butan-1-d benzeie acid and butan-2-d 2-methylbutanoic acid and phenol benzoic acid and methyl ethyl ketonearrow_forwardWhat compounds are obtained from the following hydrolysis reactions?arrow_forward
- Draw the product formed when phenylacetic acid (C6H5CH2COOH) istreated with following reagent. With some reagents, no reaction occurs. NaOHarrow_forwardThe ketone produced in the process is: H diphenylketone benzaldehyde O benzeneketone benzoyl chloride ketone AICI 3 ?arrow_forwardName the following compound: O Butyl cyclohexanoate O Butanoic cyclohexanecarboxylic anhydride O Cyclohexyl butanoate O Cyclohexanoic butanoate O Butyl cyclohexyl anhydridearrow_forward
- How might you prepare the following esters using a nucleophilic acyl substitution reaction of an acid chloride? (a) CH3CH2CO2CH3 (b) CH3CO2CH2CH3 (c) Ethyl benzoatearrow_forwardWhat is the reduction product of the following compound with H2/Pd? A 2-propyl-1-cyclohexanol B 2-propenyl-1-cyclohexanol C2-propylcyclohexanone D 1-propyl-2-cyclohexanol What is the oxidation product of this compound? H' A ethanoic acid acetic acid propanoic acid D propanone What is the oxidation product of this compound? A 3,4-dimethylpentanoic acid B 2,3-dimethylpentanoic acid C2,3-dimethylpentanone 3,4-dimethylpentanonearrow_forwardPropose an efficient synthesis for the following transformations using ONLY malonic ester synthesis.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

