
Chemistry: Principles and Practice
3rd Edition
ISBN: 9780534420123
Author: Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 22, Problem 55QE
Name the following compounds.
- (a) FCH2CH2CH2OH
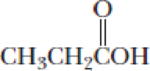
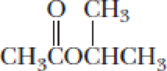
- (b) CH3CH2CH2NH2
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Draw and name compounds that meet these descriptions:(a) Three different amides with the formula C5H11NO(b) Three different esters with the formula C6H12O2
Write a condensed structural formula for each of the following:(a) an acid with the formula C4H8O2, (b) a cyclicketone with the formula C5H8O, (c) a dihydroxy compoundwith the formula C3H8O2, (d) a cyclic ester with theformula C5H8O2.
Predict the products formed in each of the following reactions, and write a balanced equation:
(a) CH3NHNH2(g) + O2(8) → ?
(b) Mg3P2(s) + H20(1) → ?
Chapter 22 Solutions
Chemistry: Principles and Practice
Ch. 22 - Prob. 1QECh. 22 -
Explain what is special about the element carbon...Ch. 22 - Prob. 3QECh. 22 - Prob. 4QECh. 22 - Prob. 5QECh. 22 - Prob. 6QECh. 22 - Prob. 7QECh. 22 - Prob. 8QECh. 22 - Prob. 9QECh. 22 - Prob. 10QE
Ch. 22 - Prob. 11QECh. 22 - Prob. 12QECh. 22 - Prob. 13QECh. 22 - Prob. 14QECh. 22 - Prob. 15QECh. 22 - Prob. 16QECh. 22 - Prob. 17QECh. 22 - Prob. 18QECh. 22 - Prob. 19QECh. 22 - Prob. 20QECh. 22 - Prob. 21QECh. 22 - Prob. 22QECh. 22 - Prob. 23QECh. 22 - Prob. 24QECh. 22 - Prob. 25QECh. 22 - Prob. 26QECh. 22 - Prob. 27QECh. 22 - Prob. 28QECh. 22 - Prob. 29QECh. 22 - Prob. 30QECh. 22 - Prob. 31QECh. 22 - Prob. 32QECh. 22 - Prob. 33QECh. 22 - Prob. 34QECh. 22 - Prob. 35QECh. 22 - Prob. 36QECh. 22 - Prob. 37QECh. 22 - Prob. 38QECh. 22 - Prob. 39QECh. 22 - Prob. 40QECh. 22 - Prob. 41QECh. 22 - Prob. 42QECh. 22 - Prob. 43QECh. 22 - Prob. 44QECh. 22 - Prob. 45QECh. 22 - Prob. 46QECh. 22 - Prob. 47QECh. 22 - Prob. 48QECh. 22 - Prob. 49QECh. 22 - Prob. 50QECh. 22 - Prob. 51QECh. 22 - Prob. 52QECh. 22 - Prob. 53QECh. 22 - Prob. 54QECh. 22 - Name the following compounds. (a) FCH2CH2CH2OH (b)...Ch. 22 - Prob. 56QECh. 22 - Prob. 57QECh. 22 - Prob. 58QECh. 22 - Prob. 59QECh. 22 - Prob. 60QECh. 22 - Prob. 61QECh. 22 - Prob. 62QECh. 22 - Prob. 63QECh. 22 - Prob. 64QECh. 22 - Prob. 65QECh. 22 - Prob. 66QECh. 22 - Prob. 67QECh. 22 - Prob. 68QECh. 22 - Prob. 69QECh. 22 - Prob. 70QECh. 22 - Prob. 71QECh. 22 - Prob. 72QECh. 22 - Prob. 73QECh. 22 - Prob. 74QECh. 22 - Prob. 75QECh. 22 - Prob. 76QECh. 22 - Prob. 77QECh. 22 - Prob. 78QECh. 22 - Prob. 79QECh. 22 - Prob. 80QECh. 22 - Prob. 81QECh. 22 - Prob. 82QECh. 22 - Prob. 83QECh. 22 - Prob. 84QECh. 22 - Prob. 85QECh. 22 - Prob. 86QECh. 22 - Prob. 87QECh. 22 - Prob. 88QE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Classify each compound (a)–(f) as one of the following: (i) amide, (ii) ester, or(iii) carboxylic acid. (a) CH3COOCH3 (b) RCONHR (c) C6H5COOHarrow_forwardDraw the condensed formula or skeletal structural formula for the following compounds: (a) 2,3-dimethylpentanoic acid (b) Ethyl pentanoate (c) N,N-dimethyl-1-propanaminearrow_forwardGive a chemical test to distinguish between each of the following pairs of compounds :(i) Ethylamine and Aniline(ii) Aniline and Benzylaminearrow_forward
- The functional group of butanone is:(a) Carboxyl(b) ketonic(c) aldehydic(d) alcoholicarrow_forwardWhich of the following describes a chemical property of methanol? (A) It is a colorless liquid; (B) It is a flammable liquid; (C) It is a volatile liquid; (D) It has a low boiling point;arrow_forwardWhat is the line bond formula and structural formula for CH2OCH2CH2CH2 ? Thank youarrow_forward
- Indicate whether each statement is true or false: (a) Fat molecules contain amide bonds. (b) Phosphoplipids can be zwitterions. (c) Phospholipids form bilayers in water in order to have their long hydrophobic tails interact favorably with each other, leaving their polar heads to the aqueous environment.arrow_forwarda) 2CH3OH Acid Catalystarrow_forwardGive the structural formulae and name the functional groups of the following compounds. (a) 3-chlorobut-1-ene Name the functional group: (b) butanedioic acid Name the functional group: (c) propanamide Name the functional group: (d) 3-methylbutanal Name the functional group:arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY