
Chemistry: The Central Science (13th Edition)
13th Edition
ISBN: 9780321910417
Author: Theodore E. Brown, H. Eugene LeMay, Bruce E. Bursten, Catherine Murphy, Patrick Woodward, Matthew E. Stoltzfus
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Textbook Question
Chapter 22, Problem 22E
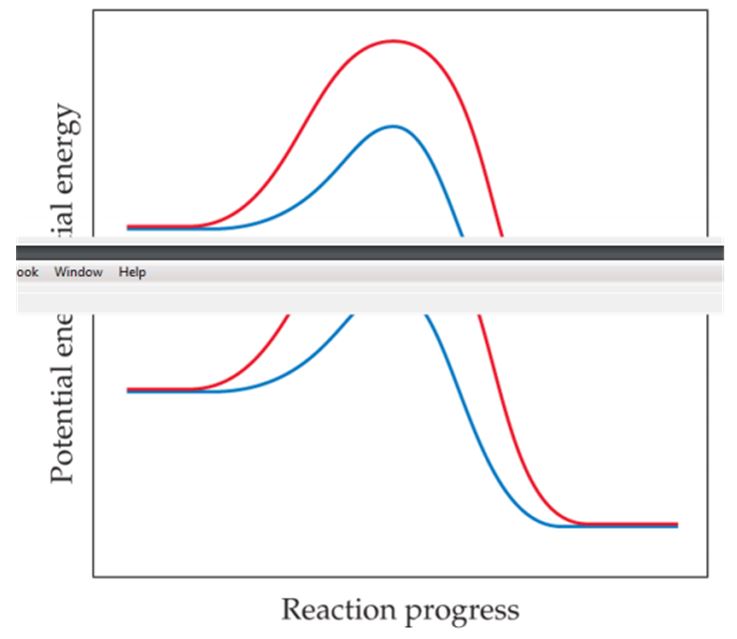
The following graph shows two different reaction pathways for the same overall reaction at the same temperature. Is each of the following statements true or false? (a) The rate is faster for the red path than for the blue path. (b) For both paths, the rate of the reverse reaction is slower than the rate of the forward reaction. (c) The energy change ΔE is the same for both paths. [Section 14.6]
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
The following graph shows two different reaction pathwaysfor the same overall reaction at the same temperature.Is each of the following statements true or false?(a) The rate is faster for the red path than for the bluepath. (b) For both paths, the rate of the reverse reactionis slower than the rate of the forward reaction. (c) The energy change ΔE is the same for both paths. [
Two gaseous compounds, A and B are introduced into an evacuated reaction chamber. The resulting reaction is exothermic and the balanced chemical equation is shown below.
A(g) + B(g) ---> C(g)
As A and B are initially introduced into the reaction chamber, which statement about the rates of the reaction is true?
(A) The rates of the forward and reverse reaction are the same.
(B) The rate of the forward reaction is faster than the rate of the reverse reaction.
(C) The rate of the forward reaction is slower than the rate of the reverse reaction.
(D) The rate cannot be determined without knowing the rate laws.
For this reaction:
Pa(s) +3 02 (g) - POg (s) + heat
Which of the following changes would INCREASE the initial rate of the reaction? More than one answer may be correct. Select all that apply.
O Grind the solid phosphorus into a fine powder
O Use more 02 (g) in the reaction mixture
Heat the reaction to a higher temperature
O Add an appropriate catalyst
Increase the volume of the reaction vessel
Hydrazine, a rocket propellant, can be made from its elements according to this reaction:
8 H2 +
3 N2
N2H4 + 4 NH3
(ammonia is also made)
When the individual rate of the H2 disappearance is -0.00795 M/s, what is the individual rate for N2?
Chapter 22 Solutions
Chemistry: The Central Science (13th Edition)
Ch. 22.1 - Prob. 21.1.1PECh. 22.1 - Prob. 21.1.2PECh. 22.1 - Prob. 21.2.1PECh. 22.1 - Prob. 21.2.2PECh. 22.3 - At 25 ° C, the decomposition of dinitrogen...Ch. 22.3 - Practice Exercise 2 The decomposition of dimethyl...Ch. 22.4 - Practice Exercise 1 For a certain reaction A ...Ch. 22.4 - Prob. 21.4.2PECh. 22.7 - Prob. 21.7.1PECh. 22.7 - Prob. 21.7.2PE
Ch. 22.10 - Prob. 21.10.1PECh. 22.10 - Prob. 21.10.2PECh. 22.10 - Prob. 21.7.1PECh. 22.10 - Prob. 21.7.2PECh. 22 - Prob. 1DECh. 22 - Prob. 1ECh. 22 - Prob. 2ECh. 22 - Prob. 3ECh. 22 - Prob. 4ECh. 22 - The gas-phase reaction CL (g) + HBr (g) + HCl (g)...Ch. 22 - What is the molecularity of each of the following...Ch. 22 - Prob. 7ECh. 22 - Prob. 8ECh. 22 - Cyclopentadiene (C5H6) reacts with itself to form...Ch. 22 - Practice Exercise 1 An Alternative two-step...Ch. 22 - Prob. 11ECh. 22 - Practice Exercise 1
Consider the...Ch. 22 - Prob. 13ECh. 22 - Prob. 14ECh. 22 - Prob. 15ECh. 22 - Prob. 16ECh. 22 - You study the rate of a reaction, measuring both...Ch. 22 - Suppose that for the reaction K+L M, you monitor...Ch. 22 - Prob. 19ECh. 22 - Prob. 20ECh. 22 - Prob. 21ECh. 22 - The following graph shows two different reaction...Ch. 22 - Prob. 23ECh. 22 - Prob. 24ECh. 22 - Prob. 25ECh. 22 - Prob. 26ECh. 22 - Prob. 27ECh. 22 - Prob. 28ECh. 22 - Prob. 29ECh. 22 - Prob. 30ECh. 22 - Prob. 31ECh. 22 - Prob. 32ECh. 22 - Prob. 33ECh. 22 - Prob. 34ECh. 22 - Prob. 35ECh. 22 - Prob. 36ECh. 22 - Prob. 37ECh. 22 - Prob. 38ECh. 22 - Prob. 39ECh. 22 - Prob. 40ECh. 22 - Prob. 41ECh. 22 - Prob. 42ECh. 22 - Prob. 43ECh. 22 - Prob. 44ECh. 22 - Prob. 45ECh. 22 - Prob. 46ECh. 22 - Prob. 47ECh. 22 - Prob. 48ECh. 22 - Prob. 49ECh. 22 - Prob. 50ECh. 22 - Prob. 51ECh. 22 - Prob. 52ECh. 22 - Prob. 53ECh. 22 - Prob. 54ECh. 22 - Prob. 55ECh. 22 - Prob. 56ECh. 22 - Prob. 57ECh. 22 - Prob. 58ECh. 22 - Prob. 59ECh. 22 - Prob. 60ECh. 22 - Prob. 61ECh. 22 - Prob. 62ECh. 22 - Prob. 63ECh. 22 - Prob. 64ECh. 22 - Prob. 65ECh. 22 - Prob. 66ECh. 22 - Prob. 67ECh. 22 - Prob. 68ECh. 22 - Prob. 69ECh. 22 - Prob. 70ECh. 22 - Prob. 71ECh. 22 - Prob. 72ECh. 22 - Prob. 73ECh. 22 - Prob. 74ECh. 22 - Prob. 75ECh. 22 - Prob. 76ECh. 22 - Prob. 77ECh. 22 - Prob. 78ECh. 22 - Prob. 79ECh. 22 - Prob. 80ECh. 22 - Prob. 81AECh. 22 - Prob. 82AECh. 22 - Prob. 83AECh. 22 - Prob. 84AECh. 22 - Prob. 85AECh. 22 - Prob. 86AECh. 22 - Prob. 87AECh. 22 - Prob. 88AECh. 22 - Prob. 89AECh. 22 - Prob. 90AECh. 22 - Prob. 91AECh. 22 - Prob. 92IECh. 22 - Prob. 93IECh. 22 - Prob. 94IECh. 22 - Prob. 95IECh. 22 - Prob. 96IECh. 22 - Prob. 97IECh. 22 - Prob. 98IECh. 22 - Prob. 99IECh. 22 - Prob. 100IECh. 22 - Prob. 101IECh. 22 - Prob. 102IECh. 22 - Prob. 103IECh. 22 - Prob. 104IECh. 22 - Prob. 105IECh. 22 - Prob. 106IECh. 22 - Prob. 107IE
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- . Plants synthesize the sugar dextrose according to the following reaction by absorbing radiant energy from the sun (photosynthesis). 6CO2(g)+6H2O(g)C6H12O6(g)+6O2(g)Will an increase in temperature tend to favor or discourage the production of C6H12O6(s)?arrow_forwardOld-fashioned smelling salts consist of ammonium carbonate, (NH4)2CO3. The reaction for the decomposition of ammonium carbonate (NH4)2CO3(s)2NH3(g)+CO(g)+H2O(g) is endothermic. Would the smell of ammonia increase or decrease as the temperature is increased?arrow_forwardThe following equation represents a reversible decomposition: CaCO3(s)CaO(s)+CO2(g) Under what conditions will decomposition in a closed container proceed to completion so that no CaCO3 remains?arrow_forward
- The direct reaction of iron(III) oxide. Fe2O3, to give iron and oxygen gas is a nonspontaneous reaction; normally, iron combines with oxygen to give rust (the oxide). Yet we do change iron(III) oxide, as iron ore, into iron metal. How is this possible? Explain.arrow_forwardConsider the reversible reaction between nitrogen and hydrogen gases shown below. N + 3H 2 (g) A In a certain experiment, nitrogen and hydrogen gases are introduced into a reaction ves- sel. Which of the following statements are true? Select all that apply. B 2 (g) C → 2NH D 3 (g) The rate of the reverse reaction increases as time goes by. Initially, only the forward reaction takes place. The rate of the forward reaction decreases as time goes by. Initially, only the reverse reaction takes place.arrow_forwardPotential Energy (kJ) 1. Consider these three reactions as the elementary steps in the mechanism for a chemical reaction. (i) Cl₂ (g) + Pt (s) → 2Cl (g) + Pt (s) AH = -950 kJ Ea = 1550 kJ Ea = 2240 kJ (ii) Cl (g)+ CO (g) + Pt (s) → CICO (g) + Pt (s) AH = 575 kJ (iii) Cl (g) + CICO (g) → Cl₂CO (g) Ea = 2350 kJ AH = -825 kJ 2600 2400 2200 2000 a. Draw the potential energy diagram for the reaction. Label the data points for clarity. The potential energy of the reactants is 600 kJ 1800 1600 1400 1200 1000 800 600 400 200 0 -200- -400 -600- -800 Reaction Progressarrow_forward
- The single-step reaction,NO2Cl(g) + NO(g) -----> NO2(g) + ONCl(g) is reversible;Ea,forward = 28.9 kJ/mol and Ea,reverse = 41.8 kJ/mol. Draw a potential energy diagram for the reaction. Indicate Ea,forward, Ea,reverse , transition state and ΔH on the diagram.arrow_forwardFor the reversible, one-step reaction, A + A k₁ K = k-1 B + C the rate constant for the forward reaction, k₁, is 265 L·mol¯¹·min¯¹ and the rate constant for the reverse reaction, k₁, is 385 L·mol¯¹·min¯¹ at a given temperature. The activation energy for the forward reaction is 42.9 kJ.mol-¹, whereas the activation energy for the reverse reaction is 23.0 kJ.mol-¹. Determine the equilibrium constant, K, of this reaction.arrow_forwardEverything needed is included. This is NOT graded, it is for practice. No grade is given for this. It is an old practice exam. The oxidation of carbon monoxide is important in reducing ground-level pollution from cars: CO(g) + 1/2 O2 (g) −−→ CO2 (g) Use the information you have available to answer the following questions. This reaction is typically run using a catalyst at elevated temperature. What is the value of ∆H◦ rxn for Equation 1 at a temperature of 700 K?arrow_forward
- 7. A general "rule of thumb" is that increasing the temperature by 10 degrees generally doubles the rate of reaction. If a reaction takes 40 seconds to occur at 45°C, specifically how long should the reaction take at the following temperatures? Explain. (a) 35⁰ (b) 55° (c) 65°arrow_forwardAnswer the following True or False. For similar reactions, the reaction with the largest value of k will be faster. F F ● ● A In general, the more concentrated the reactants are, the faster the reaction goes ● A reaction which has a rate law Rate = K[A]²[B] is third order overall. • ● A catalyst makes a reaction go faster by lowering the energy of the products. A In general, the higher the temperature, the faster the reaction goes. A/arrow_forwardEnter your answer in the provided box. The rate constant for the following elementary reaction is 6.1 x 10° M's at 25°C: -1 2A(g) +B(g) → 2C(g) AG° (kJ/mol) A 84.752 BO C 54.064 What is the rate constant for the reverse reaction at the same temperature?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
Kinetics: Chemistry's Demolition Derby - Crash Course Chemistry #32; Author: Crash Course;https://www.youtube.com/watch?v=7qOFtL3VEBc;License: Standard YouTube License, CC-BY