
Organic Chemistry (6th Edition)
6th Edition
ISBN: 9781260119107
Author: Janice Gorzynski Smith
Publisher: McGraw Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 72P
Answer the following questions about the four species
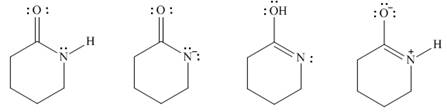
A B C D
(a) Which two species represent a conjugate acid-base pair?
(b) Which two species represent resonance structures?
(c) Which two species represent constitutional isomers?
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Answer the following questions about the four species A–D (Attached) .
Which two species represent a conjugate acid–base pair?Which two species represent resonance structures?Which two species represent constitutional isomers?
2.40 As we will see in later chapters, many steps in key reaction sequences involve acid-base reactions. (a) Draw curved arrows to
Hlustrate the flow of electrons in steps [1]-[3]. (b) Identify the base and its conjugate acid in step [1]. (c) Identify the acid and
its conjugate base in step [3].
нӧ: :ӧн
нӧ: :ӧн
:ÖH
:0:
+ H30:
+ H2ö:
HÖ.
3.
HÖ
HÖ.
Hö:
Identify the most acidic compound between the following moleculesDraw the resonance structures of the conjugate base of the acid chosen in (a). Use the curved arrows to show the movement of electrons and the resonance arrows to show the relationship between these structures. All lone electron pairs and charges must be clearly indicated
Chapter 2 Solutions
Organic Chemistry (6th Edition)
Ch. 2.1 - a. Which compounds are Bronsted-Lowry acids:...Ch. 2.2 - a. Draw the conjugate acid of each base:...Ch. 2.2 - Label each statement as True or False.
a. is the...Ch. 2.2 - Decide which compound is the acid and which is the...Ch. 2.2 - Draw the products formed from the acid-base...Ch. 2.3 - Which compound in each pair is the stronger acid?...Ch. 2.3 - Use a calculator when necessary to answer the...Ch. 2.3 - Rank the conjugate bases of each of group of acids...Ch. 2.3 - Problem-2.10 Considers two acids: (formic acid,)...Ch. 2.3 - Prob. 11P
Ch. 2.4 - Draw the products of each reaction and determine...Ch. 2.4 - Prob. 13PCh. 2.5 - Without reference to a pKa table, decide which...Ch. 2.5 - Rank the labeled H atoms in the following compound...Ch. 2.5 - Which hydrogen in pseudoephedrine, the nasal...Ch. 2.5 - Which compound in each pair is the stronger acid?...Ch. 2.5 - Glycolic acid, HOCH2CO2H, is the simplest member...Ch. 2.5 - Explain the apparent paradox. HBr is a stronger...Ch. 2.5 - The CH bond in acetone, (CH3)2C=O, has a pKa of...Ch. 2.5 - Prob. 23PCh. 2.5 - For each pair of compounds: [1] Which indicated H...Ch. 2.5 - Rank the compounds in each group in order of...Ch. 2.5 - Prob. 26PCh. 2.5 - Prob. 27PCh. 2.6 - Prob. 28PCh. 2.7 - Problem 2.29
Compounds like amphetamine that...Ch. 2.8 - Problem 2.30 Which species are Lewis bases?
a. b....Ch. 2.8 - Which species are Lewis acids?
a. b. c. d.
Ch. 2.8 - For each reaction, label the Lewis acid and base....Ch. 2.8 - Prob. 33PCh. 2.8 - Prob. 34PCh. 2.8 - Label the Lewis acid and base. Use curved arrow...Ch. 2 - 2.36 Propranolol is an antihypertensive agent—that...Ch. 2 - 2.37 Amphetamine is a powerful stimulant of the...Ch. 2 - 2.38 What is the conjugate acid of each base?
a....Ch. 2 - 2.39 What is the conjugate base of each acid?
a....Ch. 2 - Draw the products of each proton transfer...Ch. 2 - Prob. 43PCh. 2 - What is Ka for each compound? Use a calculator...Ch. 2 - What is the pKa for each compound? a. b. c.Ch. 2 - Which of the following bases are strong enough to...Ch. 2 - Draw the products of each reaction. Use the pKa...Ch. 2 - a. What is the conjugate acid of A? b. What is the...Ch. 2 - Dimethyl ether (CH3OCH3) and ethanol (CH3CH2OH)...Ch. 2 - 2.59 Atenolol is a (beta) blocker, a drug used to...Ch. 2 - 2.60 Use the principles in Section 2.5 to label...Ch. 2 - 2.61 Label the three most acidic hydrogen atoms in...Ch. 2 - Prob. 66PCh. 2 - 2.63 Classify each compound as a Lewis base, a...Ch. 2 - 2.64 Classify each species as a Lewis acid, a...Ch. 2 - Label the Lewis acid and Lewis base in each...Ch. 2 - 2.66 Draw the products of each Lewis acid-base...Ch. 2 - Prob. 71PCh. 2 - 2.68 Answer the following questions about the four...Ch. 2 - Prob. 73PCh. 2 - 2.70 Hydroxide can react as a Brønsted-Lowry base...Ch. 2 - 2.71 Answer the following questions about esmolol,...Ch. 2 - Prob. 76PCh. 2 - Prob. 77PCh. 2 - Prob. 82P
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Complete the equation for the reaction between each Lewis acid-base pair. In each equation, label which starting material is the Lewis acid and which is the Lewis base; use curved arrows to show the flow of electrons in each reaction. In doing this problem, it is essential that you show valence electrons for all atoms participating in each reaction. (a) (b) (c) (d)arrow_forwardOffer an explanation for the following observation Q.) Ethanol and water have approximately the same acidityarrow_forward1. Benzoic acid is a stronger acid than benzyl alcohol by more than ten orders of magnitude, despite both containing a hydroxyl group. Why? Draw structures to support your answer.arrow_forward
- Rank the following in order of increasing acidity. (1 = least acidic ... 4 = most acidic)arrow_forwardConsider the following reaction: (a) What is the acid on the left side of the equation? (b) What is the base on the left side of the equation? (c) What is the conjugate base of the acid on the left? (d) What is the conjugate acid of the base on the left? (e) What is the acid on the right side of the equation? (f) What is the base on the right side of the equation? (g) What is the conjugate base of the acid on the right? (h) What is the conjugate acid of the base on the right?arrow_forwardWhich is a stronger acid? Must explain with both, words and structures.arrow_forward
- Answer the following questions about the four species A–D.a.Which two species represent a conjugate acid–base pair? b. Which two species represent resonance structures? c. Which two species represent constitutional isomers?arrow_forwardWhich of the following compounds is the stronger base? Explain. or NH3 is a v base than NCI3. This is due to the difference in the electron withdrawing strength of Cl compared with H. v is more electronegative than and will v more electron density v the nitrogen than Thus, the nitrogen atom in NCI3 has a v positive charge than it does in NH3. The v positive charge means that is v difficult to add H* to NCI3 than it is to add H* to NH3. Is the compound Sb(OH)5 likely to be acidic, basic, or neutral in water? Justify your answer. Phosphorus and arsenic form acids of the form X(OH)n, so one would predict that antimony would be similar as it is in the same family. Based in the number of OH groups present, it should be However, antimony hydroxides are not stable and form oxides which are known to be amphoteric. Thus, Sb(OH)6 is in fact more likely to bearrow_forwardGiven the choices of Resonance, inductive, electronegativity, and s-character which choice best explains why the first compound is more acidic than the second one? I choose resonance factor because of the conjugate base but would s-character(hybridization ) be more important? The first compound is given to be more acidicarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY