
Concept explainers
Using the method outlined in Section
alkyl groups, and classify each one as primary, secondary, or tertiary:
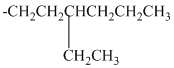
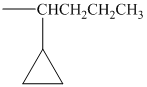
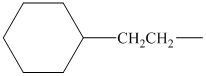
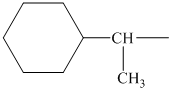
Trending nowThis is a popular solution!

Chapter 2 Solutions
Organic Chemistry - Standalone book
- 10-45 Draw structural formulas for the eight aldehydes with the molecular formula CgHi2O.arrow_forwardDodecane, C12H26, is an unbranched alkane Predict the following: Will it dissolve in water? Will it dissolve in hexane? Will it burn when ignited? Is it a liquid, solid, or gas at room temperature and atmospheric pressure? Is it more or less dense than water?arrow_forwardWhat functional group distinguishes each of the following hydrocarbon derivatives? a. halohydrocarbons b. alcohols c. ethers d. aldehydes e. ketones f. carboxylic acids g. esters h. amines Give examples of each functional group. What prefix or suffix is used to name each functional group? What are the bond angles in each? Describe the bonding in each functional group. What is the difference between a primary, secondary, and tertiary alcohol? For the functional groups in ah, when is a number required to indicate the position of the functional group? Carboxylic acids are often written as RCOOH. What does COOH indicate and what does R indicate? Aldehydes are sometimes written as RCHO. What does CHO indicate?arrow_forward
- Write the IUPAC name of the product of this reaction: HBr CH2=C(CH3)2arrow_forward4 C₂H6, C₂ H₂ and C₂ H₂. Draw their Lewis Сань 2 Structure and note the molecular geometry and the atom. bond angles around each cach Carbonarrow_forward6:35 M b N A ♥4 96% 6. Name each of the following functional groups: R— он R-0-R R-C-R R-C-N - R — с—н R-C-OHarrow_forward
- Write the systematic (IUPAC) name for each of the following organic molecules: structure OH CH3 C CH3 CH3 CH₂ CH3 CH3CH2C–CH2–CH2C−CH2CH3 CH3 CH2 OH CH3 CH3-CH₂ OH CH₂ CH3 CH3—CH,—CH–CH2–CH2–CH–CH2–CH-CH2CH3 namearrow_forward12 What type of organic molecule is this? нн | н-с-с-с H H O-H ketone O alcohol aldehyde organic acidarrow_forwardRank the compounds in order of increasing oxidation state (most reduced to most oxidized) for the carbon atom indicated in red (C*). H3C Ο Ο Ο Ο ||| < | < || || < | < ||| || < ||| < | ||| < || < | Ö: I H H3C Ö: II :O: I H₂C H H III :O Iarrow_forward
- Question 6 Answer the following questions related to the molecules whose structures are shown below. В A O H H H H H H H H H H H I I I I но-с-с-с-с-с-с-с-с-с-с-с-с-н i iiiiii i i ii H H H H H H H H H H H CH3(CH2)14-C–0–CH2–(CH2)28-CH3 O H H H H H I I I I но -с-с-с-с-с- D C HC-o0 H H H H HC-o E HC-O HC-O ofo Of the compounds in the figure, is a protective lipid and is a storage lipid. A and E OA. Cand D OB. OC, A and B OD, A and D C and E H H H H H Harrow_forward8. a) C3H$C1 has several structural isomers containing an alkene functional group. Complete the table by stating the systematic names and drawing the structural formulae of THREE structural isomers of C3H$Cl. Systematic Name Structural Formula b) A chemical test can be performed quickly in a school laboratory to distinguish between cyclohexanol and 1-methylcyclohexanol. Identify the reagent used in this test and describe the expected observations.arrow_forwardIdentify the oxygen-containing functional groups in these small molecules: H H H H H H H -C C H- H- H H H H aldehyde H H ether alcohol ketone ester aldehyde carboxylic acid ester carboxylic acid alcohol ether ketone O O O Oarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning




