
Concept explainers
(a)
Interpretation:
The structural formula for the principal product formed when benzonitrile is treated with the given reagent has to be drawn.
Concept introduction:
The reaction of
The hydrolysis reaction of alkyl nitrile in presence of acid is written as,
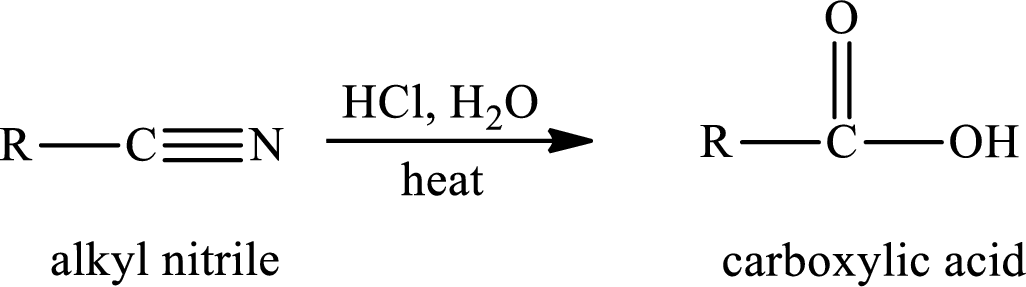
The amide is first formed when the cyano group is hydrolysed in excess of water and if there is only one mole of water per mole of nitrile, the reaction can be stopped by the formation of amide.
(b)
The structural formula for the principal product formed when benzonitrile is treated with the given reagent has to be drawn.
Concept introduction:
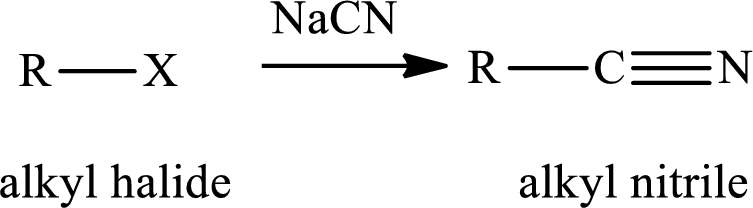
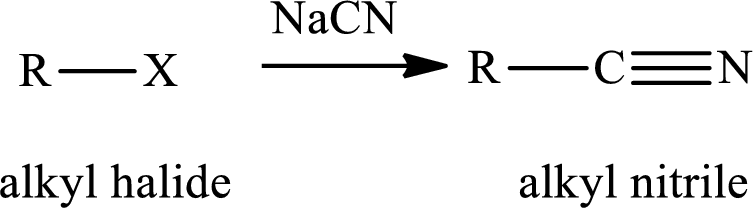
The reaction of alkyl halides with sodium cyanide gives alkyl cyanides or nitriles. After the reaction there is increase incarbon atoms of the alkyl chain of alkyl halide. The nitriles obtained undergo hydrolysis reaction in presence of acid gives carboxylic acid. The general reaction of alkyl halides and sodum cyanide is written as,
The hydrolysis reaction of alkyl nitrile in presence of acid is written as,
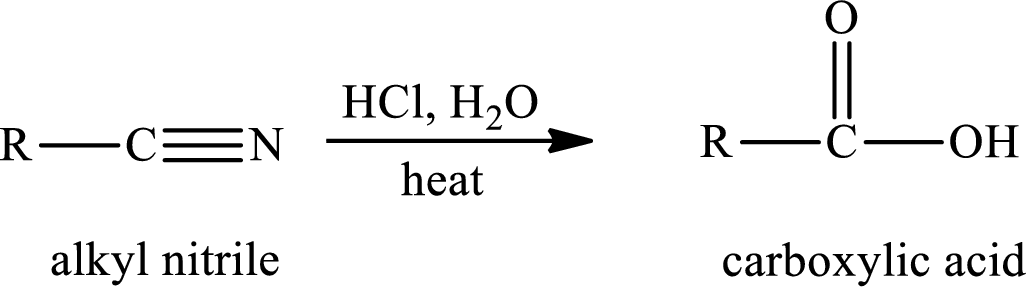
The amide is first formed when the cyano group is hydrolysed in excess of water and if there is only one mole of water per mole of nitrile, the reaction can be stopped by the formation of amide.
(c)
The structural formula for the principal product formed when benzonitrile is treated with the given reagent has to be drawn.
Concept introduction:
Amide hydrolysis in aqueous base:
A carboxylate anion and ammonia or an
The formed carboxylate anion can be converted into carboxylic acid by the acidification of the reaction mixture.
(d)
The structural formula for the principal product formed when benzonitrile is treated with the given reagent has to be drawn.
Concept introduction:
Concept introduction:
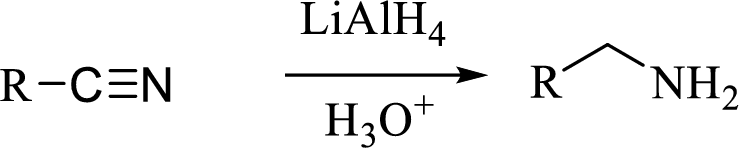
Trending nowThis is a popular solution!

Chapter 18 Solutions
Organic Chemistry
- Phenylethanol can be oxidised to phenylethanal or phenylethanoic acid, depending on the reagents used (both the alcohol and the aldehyde are of interest for their antimicrobial properties, while the acid is used to treat type II hyperammonemia): A (a) (b) (c) CoH,CH,CHO phenylethanal B C6H5CH₂CH₂OHC₂H₂CH₂CO₂H phenylethanol Suggest reagents (shown as A and B in the scheme above) that could be used to carry out the oxidation of the alcohol to the aldehyde and the acid, respectively. C6H5CH₂- Suggest two other syntheses of phenylethanoic acid, in each case indicating the starting materials and other reagents required, but not giving details of mechanism. One of your proposed syntheses must start with a compound which only contains seven carbon atoms (the acid product contains eight carbon atoms). phenylethanoic acid Phenylethanal can be converted to a hydrate in the presence of aqueous acid, though the position of equilibrium is very far to the left: H H+/H₂O OH C6H5CH₂-C-H OH Explain why…arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring. It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Part A Given 7.30 gg of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%% yield? Express your answer in grams to three significant figures. Part B A chemist ran the reaction and obtained 5.95 gg of ethyl butyrate. What was the percent yield? Express your answer as a percent to three significant figures. Part C The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0%% yield. How many grams would be produced from 7.30 gg of…arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring. It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Given 8.45 gg of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%% yield? Express your answer in grams to three significant figures. A chemist ran the reaction and obtained 5.50 gg of ethyl butyrate. What was the percent yield? Express your answer as a percent to three significant figures. The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0%% yield. How many grams would be produced from 8.45 gg of butanoic acid and excess…arrow_forward
- Ethyl butyrate, CH3CH2CH2CO2CH2CH3CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring. It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l)CH3CH2CH2CO2H(l)+CH2CH3OH(l)⟶H+CH3CH2CH2CO2CH2CH3(l)+H2O(l) A chemist ran the reaction and obtained 5.40 g of ethyl butyrate. What was the percent yield, The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 7.45g of butanoic acid and excess ethanol?arrow_forwardDibenzalpropanone is a compound that can absorb UV rays and can be used as a sunscreen. Write down the reagents used to synthesize the compound dibenzalpropanone.arrow_forwardOxalic acid is found in the leaves of rhubarb, primarilyin the form of the calcium salt. Since high levels of oxalicacid are toxic, only rhubarb stalks are used to make strawberry rhubarb pie. What is the IUPAC name of oxalic acid?Write the structure of the calcium salt of oxalic acid.arrow_forward
- Ethyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l). The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 8.50 gof butanoic acid and excess ethanol? Express your answer in grams to three significant figures.arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) a) Given 7.70 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100% yield? b) A chemist ran the reaction and obtained 5.25 g of ethyl butyrate. What was the percent yield? c) The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 7.70 g of butanoic acid and excess ethanol?arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Given 8.50 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%yield? Express your answer in grams to three significant figures.arrow_forward
- In the mid-1930s a substance was isolated from a fungus that is a parasite of ryes and other grasses. This alkaloid, lysergic acid, has been of great interest to chemists because of its strange, dramatic action on the human mind. Many derivatives of lysergic acid are known, some with medicinal applications. Perhaps the best known derivative of lysergic acid is the potent hallucinogen lysergic acid diethylamide (LSD): మగవా జి N-H LSD (CH25N;O) Like other alkaloids, LSD is a weak base, with Kp = 7.6 × 107. What is the pH of a 0.94 M solution of LSD? pH =arrow_forwardName the following carboxylic acid derivatives, giving both a common name and anIUPAC name where possible. (b) PhOCHOarrow_forwardWhich functional group has the maximum reactivity toward nucleophilic attack? a) acid chloride b) ketone c) ester d) amidearrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning



