
Concept explainers
Interpretation:
Each reacting species is to be labeled as either “electron rich” or “electron poor”. Appropriate curved arrows are to be drawn, and the type of the elementary step is to be named under the each reaction arrow for the given reaction.
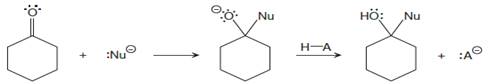
Concept introduction:
A nucleophilic addition reaction involves a
Want to see the full answer?
Check out a sample textbook solution
Chapter 17 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- Consider the following overall reaction, which will be discussed in Chapter 20.arrow_forwardShow the mechanism for the given reaction conducted at -5 °C in CCI,. cyclohexene + bromine → dibromocyclohexane Draw structures, including charges and electrons, and add curved arrows. Details count. Draw each species (organic and inorganic) resulting from the previous step. Then, add curved arrows for the forward reaction. Include charges and nonbonding electrons. Add curved arrows to the first step. : Br - Br : Draw the major product. Include charges and nonbonding electrons.arrow_forwardConsider the reaction between 2-methyl-2-butanol and HBr, shown below. но HBr Brarrow_forward
- Predict the relative rates of these reactions. That is, select 1 next to the reaction with the fastest rate, 2 next to the reaction with the next fastest rate, and so on. Note for advanced students: you may assume these reactions all take place in a polar aprotic solvent, like DMSO. ( Choose one) Predict the relative rates of these reactions. That is, select 1 next to the reaction with the fastest rate, 2 next to the reaction with the next fastest rate, and so on. Note for advanced students: you may assume these reactions all take place in a polar aprotic solvent, like DMSO. u + H₂O Reaction - OH₂ Relative Rate (Choose one) OH₂ (Choose one) ▼ +CI ... + H₂O OH + OH + I (Choose one) (Choose one) ▼ + H₂S + CIarrow_forwardDraw the products for the addition reactions below in the boxes. Then, draw the FULL electron-pushing mechanism for the reactions, including all intermediates (with formal charges and lone pairs of electrons) and all electron pushing arrows. Label the electrophile and nucleophile in each step.arrow_forwardCan you help me draw the mechanism arrows for this reaction and determine if the reaction is going forward or reverse?arrow_forward
- Would these be the correct mechanism arrows for this reaction?arrow_forwardChemistry Please also include the mechanism. Make sure to include the major products and any stereochemistry. For each arrow, there are 2 steps involved include those steps.arrow_forwardSame reaction as in Q1. H3PO4 Choose all intermediates that are formed during the reaction. Carbocation O Alkoxide Oxonium No intermediates are formedarrow_forward
- The following reaction, which is discussed in Chapter 8, is an example of a unimolecular nucleophilic substitution (Sn1) reaction. It consists of the four elementary steps shown here. For each step (i-iv), (a) identify all electron-rich sites and all electron-poor sites, (b) draw in the appropriate curved arrows to show the bond formation and bond breaking that occur, and (c) name the elementary step. (i) H-OCH3 OH + H-OCH3 H. H (ii) ©CH2 + H,O (iii) ©CH2 CH3 + HO-CH3 (iv) CH3 CH3 + H2O-CH, + НО—СНЗ foarrow_forwardIn the first box, draw the tetrahedral intermediate involved in this reaction.In the second box, draw the product of the reaction.Include all lone pairs of electrons and nonzero formal charges. Do not add curved arrows.arrow_forwardConsider the reaction between 2-methyl-2-butanol and HBr, shown below. но HBr Brarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
