
Principles of General, Organic, Biological Chemistry
2nd Edition
ISBN: 9780073511191
Author: Janice Gorzynski Smith Dr.
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Chapter 13.6, Problem 13.19P
Interpretation Introduction
Interpretation:
Products that would form when the given triacylglycerol undergoes hydrolysis in water has to be drawn.
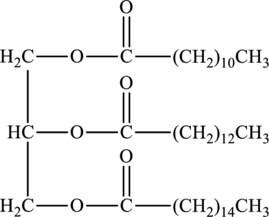
Concept Introduction:
In presence of water and acid, the esters undergo hydrolysis to form
Triacylglycerol contains three ester groups. Ester groups contains long chain hydrocarbon. Hydrolysis of triacylglycerol results in formation of three molecules of acid and one molecule of glycerol.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
What is the composition of the soap prepared by hydrolysis of the following triacylglycerol?
Draw the products formed when the given triacylglycerol is hydrolyzed with water in the presence of sulfuric acid
Is this fatty acid likely to be pro-inflammatory or anti-inflammatory?
O Pro-inflammatory
Anti-inflammatory
ONeither pro-inflammatory or anti-inflammatory
OBoth pro-inflammatory and anti-inflammatory
O
GH
Chapter 13 Solutions
Principles of General, Organic, Biological Chemistry
Ch. 13.1 - Draw out each compound to clearly show what groups...Ch. 13.1 - Prob. 13.2PCh. 13.2 - Prob. 13.3PCh. 13.2 - Give the structure corresponding to each IUPAC...Ch. 13.2 - Prob. 13.5PCh. 13.2 - Give the structure corresponding to each name. a....Ch. 13.3 - Which compound in each pair has the higher boiling...Ch. 13.3 - Rank the following compounds in order of...Ch. 13.4 - Which compounds are -hydroxy acids? tartaric acid...Ch. 13.4 - Prob. 13.10P
Ch. 13.5 - Prob. 13.11PCh. 13.5 - Prob. 13.12PCh. 13.5 - Prob. 13.13PCh. 13.6 - Prob. 13.14PCh. 13.6 - Prob. 13.15PCh. 13.6 - Prob. 13.16PCh. 13.6 - Prob. 13.17PCh. 13.6 - Prob. 13.18PCh. 13.6 - Prob. 13.19PCh. 13.7 - Prob. 13.20PCh. 13.7 - Prob. 13.21PCh. 13.7 - Prob. 13.22PCh. 13.7 - Prob. 13.23PCh. 13.7 - Prob. 13.24PCh. 13.8 - Prob. 13.25PCh. 13.8 - Prob. 13.26PCh. 13.8 - Prob. 13.27PCh. 13.8 - Draw the product formed when each ammonium salt is...Ch. 13.8 - Prob. 13.29PCh. 13.9 - Prob. 13.30PCh. 13.9 - Prob. 13.31PCh. 13.9 - Prob. 13.32PCh. 13.9 - Why is the boiling point of CH3CONH2(221C) higher...Ch. 13.9 - Prob. 13.34PCh. 13.9 - Prob. 13.35PCh. 13.10 - Prob. 13.36PCh. 13 - Prob. 13.37UKCCh. 13 - Prob. 13.38UKCCh. 13 - Prob. 13.39UKCCh. 13 - Prob. 13.40UKCCh. 13 - Prob. 13.41UKCCh. 13 - Prob. 13.42UKCCh. 13 - Prob. 13.43UKCCh. 13 - Prob. 13.44UKCCh. 13 - Prob. 13.45UKCCh. 13 - Prob. 13.46UKCCh. 13 - Prob. 13.47UKCCh. 13 - Prob. 13.48UKCCh. 13 - Prob. 13.49UKCCh. 13 - Prob. 13.50UKCCh. 13 - Prob. 13.51APCh. 13 - Prob. 13.52APCh. 13 - Prob. 13.53APCh. 13 - Draw the structure of a compound of molecular...Ch. 13 - Prob. 13.55APCh. 13 - Prob. 13.56APCh. 13 - Give an acceptable name for each compound.Ch. 13 - Prob. 13.58APCh. 13 - Prob. 13.59APCh. 13 - Prob. 13.60APCh. 13 - Prob. 13.61APCh. 13 - Prob. 13.62APCh. 13 - Prob. 13.63APCh. 13 - Give an acceptable name for each amine or amide....Ch. 13 - Draw the structure corresponding to each name. a....Ch. 13 - Draw the structure corresponding to each name. a....Ch. 13 - Prob. 13.67APCh. 13 - Draw the structure of each amine or ammonium salt....Ch. 13 - Prob. 13.69APCh. 13 - Which compound in each pair is more water soluble?...Ch. 13 - Prob. 13.71APCh. 13 - Prob. 13.72APCh. 13 - Prob. 13.73APCh. 13 - Prob. 13.74APCh. 13 - Prob. 13.75APCh. 13 - Prob. 13.76APCh. 13 - Prob. 13.77APCh. 13 - Prob. 13.78APCh. 13 - Prob. 13.79APCh. 13 - Prob. 13.80APCh. 13 - Prob. 13.81APCh. 13 - Prob. 13.82APCh. 13 - Prob. 13.83APCh. 13 - Prob. 13.84APCh. 13 - Prob. 13.85APCh. 13 - Prob. 13.86APCh. 13 - Prob. 13.87APCh. 13 - Draw the products of each acid-base reaction.Ch. 13 - Prob. 13.89APCh. 13 - Prob. 13.90APCh. 13 - Prob. 13.91APCh. 13 - Prob. 13.92APCh. 13 - Ritalin is the trade name for methylphenidate, a...Ch. 13 - Prob. 13.94APCh. 13 - Prob. 13.95CPCh. 13 - Prob. 13.96CPCh. 13 - Prob. 13.97CPCh. 13 - Prob. 13.98BTCCh. 13 - Prob. 13.99BTCCh. 13 - Prob. 13.100BTC
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- What additional products are formed when a triacyl-glyoerol molecule is converted to each of the following in an acidic hydrolysis reaction. a. diacylglycerol b. monoacylglycerol c. glycerolarrow_forwardClassify each of the following as (1) an energy-storage lipid, (2) a membrane lipid, or (3) an emulsification lipid. a. Oils b. Glycocholic acid c. Glycerophospholipids d. Sphingophospholipidsarrow_forwardDraw condensed structural formulas for all products you would obtain from the saponification with KOH of the triacylglycerol in Problem 19-54.arrow_forward
- Draw condensed structural formulas for all products you would obtain from the saponification with NaOH of the triacylglycerol in Problem 19-53.arrow_forwardDraw the structure of a triacylglycerol formed from glycerol, one molecule of stearic acid, and two molecules of oleic acid. The stearic acid should form an ester bond with the 2° OH group of glycerol.arrow_forwardIn addition to glycerol, fatty acids and phosphate, phosphoglycerols generally contain: nonpolar amino acids creatine O polar or ionic alcohols O sterols O aromatic amino acidsarrow_forward
- of the triacylglyceról 19-56 Draw condensed structural formulas for all products you would obtain from the saponification with KOH of the triacylglycerol in Problem 19-54.arrow_forwardIdentify the polar and nonpolar regions in the following fatty acid: но Is the fatty acid saturated or unsaturated? Is the fatty acid an omega 3, omega 6 or neither?arrow_forwardDraw the products formed when triacylglycerol A is treated with each reagent. Rank compounds A, B, and C in order of increasing melting point.arrow_forward
- How many molecules of H2 will react with one molecule of the following triacylglycerol? CH2- CH-O O=U O=Uarrow_forwardWhich of the following is an unsaturated triacylglycerol? 요 0 FO 0- =ㅁ 아 -NH5arrow_forwardWhat is the composition of the soap prepared by hydrolysis of the attachedtriacylglycerol?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,

General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

Organic And Biological Chemistry
Chemistry
ISBN:9781305081079
Author:STOKER, H. Stephen (howard Stephen)
Publisher:Cengage Learning,
Lipids - Fatty Acids, Triglycerides, Phospholipids, Terpenes, Waxes, Eicosanoids; Author: The Organic Chemistry Tutor;https://www.youtube.com/watch?v=7dmoH5dAvpY;License: Standard YouTube License, CC-BY