
Concept explainers
Interpretation:
The missing reagents for each step in Your Turn 13.6 are to be supplied.
Concept introduction:
In order to identify the missing reagents in the given reaction sequence, it is important to identify if the reaction involves a
Under basic conditions, the nucleophile attacks the
Answer to Problem 13.1P
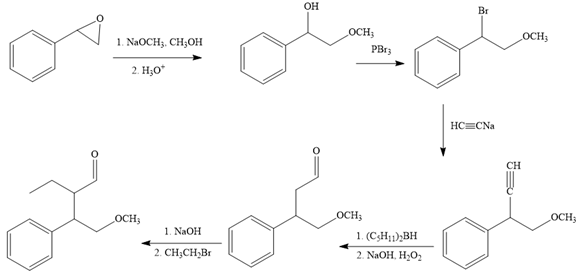
The missing reagents for each step in the given reaction sequence are given below:
Explanation of Solution
The reaction sequence given in Your Turn 13.6 is:
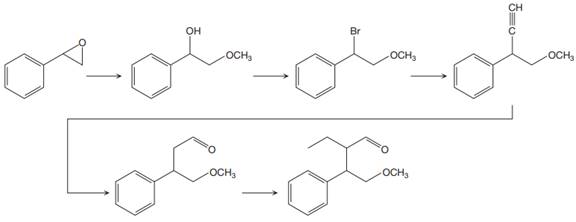
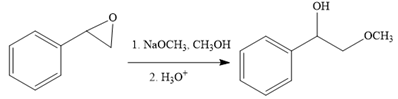
The first reaction is the conversion of an epoxide to alcohol. Thus, it is a functional group transformation reaction in which the nucleophile,
The first reaction and the missing reagents for it are shown below:
The second reaction also involves functional group transformation. The alcoholic (
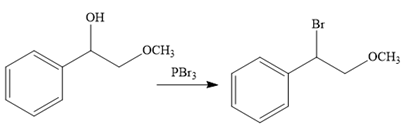
In the third reaction, the bromine atom is replaced by an acetylene group (
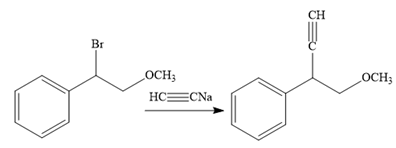
The fourth reaction in the given reaction sequence is a reaction involving a functional group transformation. Terminal alkynes undergo a hydroboration-oxidation reaction which leads to the formation of an aldehyde. The reagents used in the hydroboration-oxidation reaction are disiamylborane [
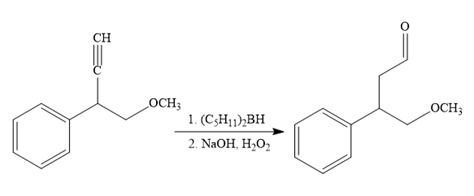
The fifth reaction involves the alteration of the carbon skeleton. The alpha hydrogen attached to an alpha carbon in aldehydes is weakly acidic. A strong base abstracts this alpha hydrogen to form an enolate ion. This enolate serves as a nucleophile and reacts with an alkyl halide via
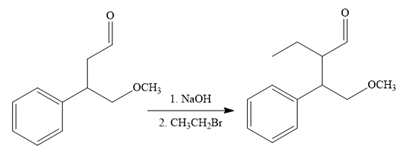
The complete reaction sequence with appropriate reagents for each are given below:
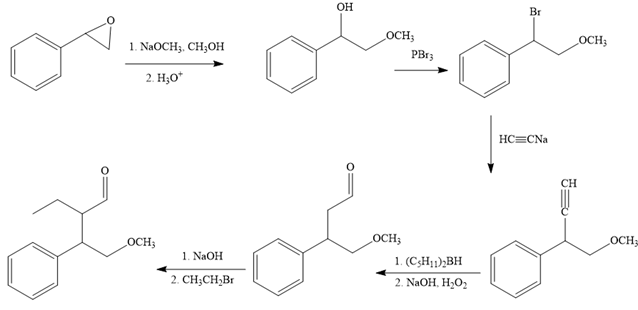
In order to identify the missing reagents in the given reaction sequence, it is important to identify if the reaction involves a functional group transformation or it is a reaction that alters the carbon skeleton.
Want to see more full solutions like this?
Chapter 13 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- What reagent(s) can be used for that last reaction. Only use ones available in the answer bank please.arrow_forwardConsider this nucleophilic substitution reaction. 1. Highlight the electrophilic carbon in red, and highlight the leaving group in blue. Highlight the atom in the nucleophile that will attack the electrophilic center in green. Only atoms need to be highlighted and not the lone pairs or formal charges. 2. Draw the product(s) of the reaction. Include all lone pairs.arrow_forwardComplete each reaction by putting the missing reagent or missing product.arrow_forward
- This reaction is an example of conjugate addition of a nucleophile to an a,ẞ-unsaturated carbonyl. H3C LOCH3 H₂O H3C OCH3 OCH3 Draw the two resonance structures of the enolate anion intermediate for this reaction. • Draw an R1 group in place of CoA. The R group tool is located in the charges and lone pairs drop-down menu. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corner. Separate resonance structures using the symbol from the drop-down menu. • O H CH3 ? [Farrow_forwardDetermine the steps to proceed the following synthesis. Please analyze the 4th reaction.arrow_forwardUse the References to access important values if needed for this question. Complete the following reaction by writing the name of the product. Ni or Pt + H2 CH3CH2CH2CH2C-H Submit Answer Retry Entire Group 1 more group attempt remai Previous Nextarrow_forward
- Use Figure 2 and Figure 3 to explain if you made a pure product or had unreacted reagents. First Picture - shows figure 2 and 3 Secons Picture shows my own results for the product to compare with.arrow_forward1. Draw the starting structure that would lead to this major product 2. Draw the major product of this reaction. Ignore inorganic 3. Draw the major product of this reaction. Ignore inorganic under these conditions. byproducts. byproducts. H3O* H3O* H3O* НОarrow_forwardCircle the correct bolded word. a. Hydrogenation of an alkyne with palladium on carbon can / cannot be controlled to give an alkene. b. When a reaction becomes more exothermic, the transition state looks more like the substrate / product. C. Addition of two equivalents of HBr to an alkyne results in a vicinal / geminal dibromide. d. An SN2 reaction involves the collision of two alkyl halides to form a new carbon-carbon bond. True / Falsearrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
