
Concept explainers
What are the major IR absorptions in the
a.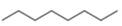 d.
d.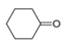
b.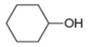
c. 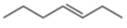 e.
e. 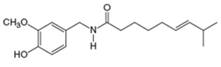
capsaicin
(spicy component of hot peppers)
(a)
Interpretation: The major IR absorptions in the functional group region for the given compound are to be predicted.
Concept introduction: IR spectroscopy is used to identify the functional group present in a compound. Each and every bond vibrates at a characteristic frequency.
Answer to Problem 13.19P
The major IR absorption peaks are observed for
Explanation of Solution
The given compound is octane. It contains
The major IR absorption peaks are observed for
(b)
Interpretation: The major IR absorptions in the functional group region for the given compound are to be predicted.
Concept introduction: IR spectroscopy is used to identify the functional group present in a compound. Each and every bond vibrates at a characteristic frequency.
Answer to Problem 13.19P
The major IR absorptions are observed for
Explanation of Solution
The given compound is cyclohexanol. It contains
The major IR absorptions are observed for
(c)
Interpretation: The major IR absorptions in the functional group region for the given compound are to be predicted.
Concept introduction: IR spectroscopy is used to identify the functional group present in a compound. Each and every bond vibrates at a characteristic frequency.
Answer to Problem 13.19P
The major IR absorptions are observed for
Explanation of Solution
The given compound is hept-3-ene. It contains
The major IR absorptions are observed for
(d)
Interpretation: The major IR absorptions in the functional group region for the given compound are to be predicted.
Concept introduction: IR spectroscopy is used to identify the functional group present in a compound. Each and every bond vibrates at a characteristic frequency.
Answer to Problem 13.19P
The major IR absorptions are observed for
Explanation of Solution
The given compound is cyclohexanone. It contains
The major IR absorptions are observed for
(e)
Interpretation: The major IR absorptions in the functional group region for the given compound are to be predicted.
Concept introduction: IR spectroscopy is used to identify the functional group present in a compound. Each and every bond vibrates at a characteristic frequency
Answer to Problem 13.19P
The major IR absorptions are observed for
Explanation of Solution
The given compound is capsaicin as shown below.
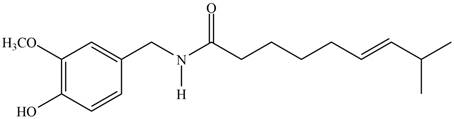
Figure 1
It contains
The major IR absorptions are observed for
Want to see more full solutions like this?
Chapter 13 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Chemistry
Essential Organic Chemistry (3rd Edition)
Organic Chemistry
Inorganic Chemistry
Introduction to Chemistry
General, Organic, & Biological Chemistry
- Which compound has the stretching vibration for its carbonyl group at the highest frequency: a.acetyl chloride, methyl acetate, or acetamide? b. Which one has the stretching vibration for its carbonyl group at the lowest frequency?arrow_forwardB.21 How would each of the following pairs of compounds differ in their IR spectra? a. b. C. d. and OH OCH 3 and -OCH3 and and OCH3 OCH₂ ENarrow_forward(A) Using only IR Spectroscopy, how would you determine the difference between the constitutional isomers listed below? a. b. C. d. a. b. C. and d. and (B) Using only ¹H - NMR, how would you determine the difference between the constitutional isomers below? OH and شما and and and and and OH HO OHarrow_forward
- a. Which compound has the stretching vibration for its carbonyl group at the highest frequency: acetyl chloride, methyl acetate, or acetamide?b. Which one has the stretching vibration for its carbonyl group at the lowest frequency?arrow_forwardWhat major IR absorptions are present above 1500 cm- for each compound? а. b.arrow_forwardThe 'H NMR chemical shifts of nitromethane, dinitromethane, and trinitromethane are at 8 6.10, 8 4.33, and 8 7.52. Match each chemical shift with the compound. Explain how chemical shift correlates with pK.arrow_forward
- Rank the four groups (a-d) from highest to lowest priority using Cahn-Ingold-Prelog rules. a d H ABCD BDCA BCDA CBDA -F Carrow_forwardExplain how you would distinguish between the following set of compounds using ¹H NMR. A. B. Number of ¹H NMR signals in Compound A: Number of ¹H NMR signals in Compound B: 6 4 12 Inarrow_forwardDiethyl ether is effective at preventing the fading of the endpoint. Explain why fading takes place and why diethyl ether is effective at preventing it.arrow_forward
- 16.64 Which of the two 13C NMR spectra below corresponds to chlorocyclohexane and which corresponds to iodocyclohexane? Explain. 200 150 100 50 200 150 100 50 Chemical shift (ppm) Chemical shift (ppm)arrow_forwardSaquinavir (trade name Incirase) belongs to a class of drugs called protease inhibitors, which are used to treat HIV. Locate all the stereogenic centers in the drug saquinavir. H. H. N. N. H. NH2 OH O: saquinavir Trade name: Invirase NH O:arrow_forwardWhich carbon would have a signal farthest downfield in the 13C NMR spectrum for this molecule? d. CI H. C. a O A. a B. b OD.d E. earrow_forward
