
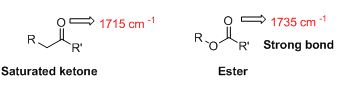
Interpretation:
The strongest bond has to be identified from C=O bond in an ester (1735 cm-1) or the C=O bond in a saturated
Concept introduction:
Spectroscopy:
It is study of the interaction of matter and
IR frequency (cm-1):
It is the number of wave crests that pass by a given point in one second frequency has units of hertz (Hz).
Stretch vibrations:
It is a vibration occurring along the line of the bond a stretching vibration changes the bond length.
Bending vibrations:
It is a vibration that does not occur along the line of the bond, bending vibration changes the bond angle.
Given information:
The IR value is given below,
Trending nowThis is a popular solution!

Chapter 12 Solutions
Organic Chemistry
- Convert neopentane (molecule A) to 4,4-dimethylpentanal (molecle B):arrow_forwardDraw a structural formula for the substitution product of the reaction shown below. H/ ● (CH3)3C |YY Br ΔΟ H Na • Use the wedge/hash bond tools to indicate stereochemistry where it exists. • If more than one stereoisomer of product is formed, draw both. Separate multiple products using the + sign from the drop-down menu. • Products that are initially formed as ions should be drawn in their neutral forms. SH acetone [Farrow_forwardCyclic forms of D-glucose: Furanose and Pyranose Recall that aldehyde and alcohol react to form hemiacetal. The O in OH acts as nucleophile, attacking the partially positive C of C=O. The H from the OH of alcohol is transferred to the O of C=O. он R R1 HO, On the other hand, ketone and alcohol also react to form hemiketal. OH R R2 R R1 HO, In glucose the OH in C-4 or C-5 can react with the C=O. If the C-4 OH reacts with the C=O the structure is a furanose (5-membered ring); if C-5 OH the product is a pyranose (6-membered ring). Furanose and pyranose are derived from cyclic ethers furan and pyran. Both furanose and pyranose have two isomers: the alpha and beta (also called anomers). Furan Pyran Furanose and pyranose form of D-glucose (note the numbering of carbon atoms) Furanose (5-membered ring) Alpha Pyranose (6-membered ring) Alpha Beta Beta 1 HO- Но H- Но H- _2 FH- H- OH FHO- H HO. но- H- но- H. Но Но H-4 H_5. FHO- FHO- H OH 6CH2OH H CH,OH CH,OH CH2OH C-1 OH is on the right Alpha-D-…arrow_forward
- Which of the following compounds does NOT absorb in the UV-visible range (200 - 900 nm)? Select one: Phenol Hexane 1,4- Benzoquinone Pentan-1,4-dien-3-one[CH2=CHC(=O)CH=CH2] Benzenearrow_forwardExample: H R 11' COO- Ca ↓ NH3 L or D?arrow_forwardBelow is a structural formula. What is the corresponding condensed structural H formula? H1C1I Н Н OCH3NHC(CH3)2 H-CN-C-C CH3NHCH(CH3)2 OCH3NC(CH3)2 :Z―I OCH3NCH(CH3)2 H H H I— H -C-H H (arrow_forward
- Fill in the table with the partial charges of the atoms. The atoms are numbered with the following scheme with respect to the substituent on the aromatic ring. x c/ 3. | H./ Atom Chlorobenzene Nitrobenzene Aminobenzene C2 Ca Which substituent results in a ring atom with the greatest positive charge (smallest negative charges)? What is the mechanism that causes this pattern of electron distribution on the aromatic ring? Diagram the mechanism. Which substituent results in a ring atom with the least positive charge (largest negative charges)? What is the mechanism that causes this pattern of electron distribution on the aromatic ring? Diagram the mechanism.arrow_forwardDraw a resonance structure, complete with all formal charges and lone (unshared) electron pairs, that shows the resonance interaction of the acetoxy with the para position in phenyl acetate. phenyl acetate ● CH3 • You do not have to consider stereochemistry. • Include all valence lone pairs in your answer. In cases where there is more than one answer, just draw one. Sn [F ?arrow_forwardGiven the following table of bond dissociation energies (in kcal/mol), what is AH° for this reaction? (CH3);С —Н + Br -Br (CH);С — Br + Н—Br | CH3-H 105 (CH3)3C-H 91 CH3-Br 70 (CH3)3C-Br 65 Br-Br 46 H-Br 88 O +7 kcal/mol O -7 kcal/mol O +16 kcal/mol O -16 kcal/mol O cannot be determined with this informationarrow_forward
- N,N-diethyl-m-toluamide (DEET) is the active ingredient in many insect repellent preparations. Following is one of the steps in its synthesis. In the box below draw the structure of the product of this reaction. MgBr H3C 0 ▾ + 1. CO₂ 2. H3O+ • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. • Draw the Grignard reagent as a covalent magnesium bromide. n [ ]# product ChemDoodleⓇ 28arrow_forwardDraw a structural formula for the major organic product of the reaction shown below. C(CH3)3 CH2 + ether H3O + CuLi H3C 2 • You do not have to consider stereochemistry. + - 4 √n [ ? ChemDoodle Ⓡarrow_forwardCH3 NaH Br Compound F CeHgO St. CH3 Propose a structure for compound F and the product C6H3O. • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • Do not include lone pairs in your answer. They will not be considered in the grading. • Draw organic products only. Do not include counter-ions, e.g., Na, I, in your answer. • Draw one structure per sketcher. Add additional sketchers using the drop-down menu in the bottom right corn • Separate structures using the sign from the drop-down menu. opy astearrow_forward
