
Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN: 9780618974122
Author: Andrei Straumanis
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 7E
Label each atom marked with an arrow with the appropriate shape name, and estimate the bond angles around it as being close to one of 180°, 120°, 109.5° or 90°. (circled charges indicate the charge on the molecule or fragment)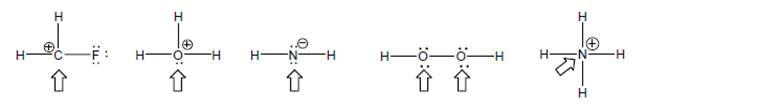
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
How do you know when to draw a solid wedge vs a dashed wedge when drawing 3D bond-line structures? I know that solid-wedge means the atom is pointing towards you and dashed wedge means it's in the back, but how do you know which atoms are in the front as opposed to the back? How can you tell what the configuration will look like in space just by looking at the lewis structure or name?
Draw the resonance structure indicated by the curved arrows. Assign formal charges.
H
H/:O:
H
H-C-C-C-Ċ—H
H H
H
Draw the molecule by placing atoms on the canvas and connecting them with bonds. Include all hydrogen atoms and nonbonding electrons. Show the formal charges of
all atoms in the correct structure.
Based on the atom connectivity shown bellow,evaluate the four resonance structure for the thiosulfate ion S2O3 ^2-.
Use curved arrows to indicate how you get from one resonance structure to another. Assign formal changes to all atoms and determine which of these resonance structure is the most stable based on a formal charge analysis
Explain your answer thoroughly. Look at the picture.
Chapter 1 Solutions
Organic Chemistry: A Guided Inquiry
Ch. 1 - (E) What does the number (+Z) at the center of...Ch. 1 - Prob. 2CTQCh. 1 - Prob. 3CTQCh. 1 - Prob. 4CTQCh. 1 - Prob. 5CTQCh. 1 - Prob. 6CTQCh. 1 - Prob. 7CTQCh. 1 - You hear a student from a nearby group say that...Ch. 1 - Use VSEPR to explain why the HBH bond angle of BH3...Ch. 1 - Both the HCH and HCO bond angles of H2CO...
Ch. 1 - Prob. 11CTQCh. 1 - Consider the following flat drawing of methane...Ch. 1 - Use VSEPR to assign a value of (close to) 109.5,...Ch. 1 - A student draws the picture of ammonia (NH3) in...Ch. 1 - Prob. 15CTQCh. 1 - How many central atoms does the molecule H2NCH3...Ch. 1 - Indicate the bond angle and shape about each...Ch. 1 - Explain how there can be two kinds of bent:...Ch. 1 - A student makes the following statement: “The...Ch. 1 - A student who missed this class needs to know how...Ch. 1 - Prob. 1ECh. 1 - Prob. 2ECh. 1 - Consider the incomplete valence shell...Ch. 1 - How many valence electrons does a neutral a. K...Ch. 1 - Consider the molecules AlCl3 (aluminum chloride)...Ch. 1 - Draw an example of a bent molecule with a bond...Ch. 1 - Label each atom marked with an arrow with the...Ch. 1 - a model of each of the following molecules: a....
Additional Science Textbook Solutions
Find more solutions based on key concepts
4.1 Write the symbols for the following elements.
a. copper
b. platinum
c. calcium
d. manganese
e. Iron
...
Chemistry: An Introduction to General, Organic, and Biological Chemistry (13th Edition)
The structural formula of 1, 2-dimethylbenzene needs to be drawn. Concept introduction: The ring structures of ...
Chemistry: Matter and Change
The method to determine the volume of a powered solid, liquid and a rock needs to be determined. Concept introd...
Living By Chemistry: First Edition Textbook
Q1. What is the empirical formula of a compound with the molecular formula
Chemistry: A Molecular Approach
Practice Exercise 1
Which of the following factors determines the size of an atom? a. the volume of the nucleus...
Chemistry: The Central Science (13th Edition)
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw two resonance structures of the given molecule/ion. Be sure to include all lone pairs. :Ö:arrow_forwardWhat is the structural diagram of CH2ClF without lone pairs? Also what is its Electronegativity bond after you subtract the smaller from the greater value along with its bond type, total lone pairs of electrons and total bonding pairs of electrons? Lastly, is it polar or non polar (polar molecularity) ?arrow_forwardA resonance hybrid is a structure that can be depicted by more than one valid Lewis structure. part1: Draw the major resonance form of fulminic acid, HCNO, with the atoms connected as indicated in the formula. Your structure should have nonzero formal charges minimized, and it should include all nonzero formal charges and all nonbonding electrons. part2: Draw the second most important resonance form of fulminic acid, HCNO, with the atoms connected as indicated in the formula. Your structure should have nonzero formal charges minimized, and it should include all nonzero formal charges and all nonbonding electrons. part3: Draw the least important resonance contributor for fulminic acid, HCNO, with the atoms connected as indicated in the formula. Your structure should have nonzero formal charges minimized and should include all nonzero formal charges and all nonbonding electrons.arrow_forward
- A resonance hybrid is a structure that can be depicted by more than one valid Lewis structure. part1: Draw the major resonance form of fulminic acid, HCNO, with the atoms connected as indicated in the formula. Your structure should have nonzero formal charges minimized, and it should include all nonzero formal charges and all nonbonding electrons. part2: Draw the second most important resonance form of fulminic acid, HCNO, with the atoms connected as indicated in the formula. Your structure should have nonzero formal charges minimized, and it should include all nonzero formal charges and all nonbonding electrons.arrow_forward< Complete the following structural formula for a neutral molecule by adding H atoms to complete the valence of each atom. Do not introduce any double or triple bonds. Then complete the Lewis diagram by adding any unshared electron pairs needed, so that each atom except H has a complete octet. [Review Topics] [References] Use the References to access important values if needed for this question. Br Br C—C— Write the molecular formula in the order CHX, where X stands for Cl or Br. Submit Answer The number of unshared pairs in the Lewis diagram unshared pair(s). Retry Entire Group 9 more group attempts remaining Previous Email Instructor Next Save and Earrow_forwardDraw the shapes of the following molecules and ions in 3-dimension. Show clearly any lone pairs of electrons on the central atom, state the number of bond pairs and lone pairs of electrons on the central atom and name the shape of the molecule or ion. (a) SiCI4, silicon tetrachloride (b) PBr3, phosphorus tribromide (c) CI2O, dichlorine oxide Provide everything stated in the instructions for each compound.arrow_forward
- Unshared, or lone, electron pairs play an important role in determining the chemical and physical properties of organic compounds. Thus, it is important to know which atoms carry unshared pairs. Use the structural formulas below to determine the number of unshared pairs at each designated atom. Be sure your answers are consistent with the formal charges on the formulas. H3C H₂C a O ao H₂ b b Hc N CH₂ Submit Answer OC₂H5 The number of unshared pairs at atom a is The number of unshared pairs at atom bis The number of unshared pairs at atom cis The number of unshared pairs at atom a is The number of unshared pairs at atom b is The number of unshared pairs at atom cis Retry Entire Group 9 more group attempts remaining V 0 1 2 3 4 Previous Email Instructor Next Save and Exitarrow_forwardSome of these structural formulas are incorrect (i.e., they do not represent a real compound) because they have atoms with an incorrect number of bonds. Which structural formulas are incorrect? Which atoms in them have an incorrect number of bonds?arrow_forwardUnshared, or lone, electron pairs play an important role in determining the chemical and physical properties of organic compounds. Thus, It Is Important to know which atoms carry unshared pairs. Use the structural formulas below to determine the number of unshared pairs at each designated atom. Be sure your answers are consistent with the formal charges on the formulas. H₂C O H₂ b CH₂ C Hc H₂C-CH₂ Submit Answer The number of unshared pairs at atom a is The number of unshared pairs at atom b is The number of unshared pairs at atom c is The number of unshared pairs at atom a is The number of unshared pairs at atom b is The number of unshared pairs at atom c is Retry Entire Group 9 more group attempts remaining Previous Email Instructor Next Save and Exitarrow_forward
- Draw the Lewis structures for the following four molecules, being sure to show all steps following the methods covered in class. Structures without work shown will be marked incorrect. Also, one of these molecules has resonance structures – for this compound, make sure to include all resonance structures, indicate formal charges for each atom. SO2 OF2 IF3 NH4+arrow_forwardProvide the formal charges for each atom in the molecule below, and answer the additional question (hint: none violate the octet rule). Format your answer as +2 or -3, for example. If there is no formal charge, then enter a zer SECIN S: s this molecule have an overall charge (yes or no)? harrow_forwardDraw the Lewis structures for the following four molecules, being sure to show all steps following the methods covered in class. Structures without work shown will be marked incorrect. Also, one of these molecules has resonance structures – for this compound, make sure to include all resonance structures, indicate formal charges for each atom. SO2 OF2 IF3 NH4+ Consider a molecule where the central atom has one lone pair of electrons and is double-bonded to two other atoms (of a different element). Draw a general diagram of the molecule. Is this molecule likely a polar or nonpolar molecule? Briefly explain your reasoning, using words and the diagram.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

Stoichiometry - Chemistry for Massive Creatures: Crash Course Chemistry #6; Author: Crash Course;https://www.youtube.com/watch?v=UL1jmJaUkaQ;License: Standard YouTube License, CC-BY
Bonding (Ionic, Covalent & Metallic) - GCSE Chemistry; Author: Science Shorts;https://www.youtube.com/watch?v=p9MA6Od-zBA;License: Standard YouTube License, CC-BY
General Chemistry 1A. Lecture 12. Two Theories of Bonding.; Author: UCI Open;https://www.youtube.com/watch?v=dLTlL9Z1bh0;License: CC-BY