
Concept explainers
(a)
Interpretation:The overlap of orbital that are used to form every bond with indicated atom in the given molecule needs to be drawn.
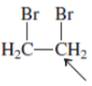
Concept Introduction: Hybridization involves the mixing of atomic orbitals to form same number of hybrid orbitals. These hybrid orbitals overlap with atomic orbital of other atoms to form covalent bond. These hybrid orbitals are of same energy and share therefore overlap effectively to form covalent bond.
The hybridization gives idea about the geometry of each atom. It can be checked with the below formula:
Hybridization = Number of sigma bonds + Number of lone pairs on bonded atoms.
(b)
Interpretation: The overlap of orbital that are used to form every bond with indicated atom in the given molecule needs to be drawn.
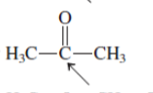
Concept Introduction: Hybridization involves the mixing of atomic orbitals to form same number of hybrid orbitals. These hybrid orbitals overlap with atomic orbital of other atoms to form covalent bond. These hybrid orbitals are of same energy and share therefore overlap effectively to form covalent bond.
The hybridization gives idea about the geometry of each atom. It can be checked with the below formula:
Hybridization = Number of sigma bonds + Number of lone pairs on bonded atoms.
(c)
Interpretation: The overlap of orbital that are used to form every bond with indicated atom in the given molecule needs to be drawn.
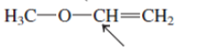
Concept Introduction: Hybridization involves the mixing of atomic orbitals to form same number of hybrid orbitals. These hybrid orbitals overlap with atomic orbital of other atoms to form covalent bond. These hybrid orbitals are of same energy and share therefore overlap effectively to form covalent bond.
The hybridization gives idea about the geometry of each atom. It can be checked with the below formula:
Hybridization = Number of sigma bonds + Number of lone pairs on bonded atoms.
(d)
Interpretation: The overlap of orbital that are used to form every bond with indicated atom in the given molecule needs to be drawn.
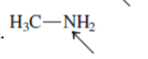
Concept Introduction: Hybridization involves the mixing of atomic orbitals to form same number of hybrid orbitals. These hybrid orbitals overlap with atomic orbital of other atoms to form covalent bond. These hybrid orbitals are of same energy and share therefore overlap effectively to form covalent bond.
The hybridization gives idea about the geometry of each atom. It can be checked with the below formula:
Hybridization = Number of sigma bonds + Number of lone pairs on bonded atoms.
(e)
Interpretation: The overlap of orbital that are used to form every bond with indicated atom in the given molecule needs to be drawn.
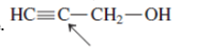
Concept Introduction: Hybridization involves the mixing of atomic orbitals to form same number of hybrid orbitals. These hybrid orbitals overlap with atomic orbital of other atoms to form covalent bond. These hybrid orbitals are of same energy and share therefore overlap effectively to form covalent bond.
The hybridization gives idea about the geometry of each atom. It can be checked with the below formula:
Hybridization = Number of sigma bonds + Number of lone pairs on bonded atoms.
(f)
Interpretation: The overlap of orbital that are used to form every bond with indicated atom in the given molecule needs to be drawn.
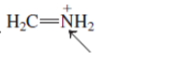
Concept Introduction: Hybridization involves the mixing of atomic orbitals to form same number of hybrid orbitals. These hybrid orbitals overlap with atomic orbital of other atoms to form covalent bond. These hybrid orbitals are of same energy and share therefore overlap effectively to form covalent bond.
The hybridization gives idea about the geometry of each atom. It can be checked with the below formula:
Hybridization = Number of sigma bonds + Number of lone pairs on bonded atoms.
Trending nowThis is a popular solution!

Chapter 1 Solutions
Organic Chemistry: Structure and Function
- Part G. Molecules with Oxygen and Nitrogen. Organic molecules often contain the elements oxygen and nitrogen. Construct a model with the molecular formula C5H12O where the oxygen is bonded to one hydrogen. When a hydrogen is bonded to an oxygen or nitrogen, you need to draw that bond in a bond-line structure. Draw the bond-line structure of this model. What is the molecular geometry of oxygen in your structure? Draw three more constitutional isomers with the molecular formula C5H12O where the oxygen is bonded to one hydrogen. Structure Structure Structure Draw three more constitutional isomers with the molecular formula C5H12O where the oxygen is bonded to two carbon atoms. Structure Structure Structurearrow_forwardWhat orbitals are used to form each highlighted bond? For multiple bonds, indicate the orbitals used in individual bonds.arrow_forwardIncorrect. This bond is made of an sp3 orbital from a carbon atom to an sp orbital on the other carbon, containing the triple bond. The correct orbital overlap is sp3-sp. What type(s) of orbital overlap is(are) indicated on the following structure:arrow_forward
- draw diagram that best describes the orbitals involving non-hydrogen bonds in C H3 O H?arrow_forwardWhich of these statements concerning p-orbitals is false? 1. They consist of two equivalent lobes. 2. They are absent from the first shell of atomic orbitals. 3. They can form & bonds. 4. They only participate in bonding on carbon atoms. 5. They can hold a maximum of two electrons.arrow_forwardSee the Attached compound A. Ques: Label the weakest C–C bond.arrow_forward
- How many of the following molecules will rotate plane polarized light? H. CH, CH3 Me H. CH, CH,CH3 H;C. CH2CH3 H;C. H. ČH, Br cı' H H;CH,C 2arrow_forward1. Is the following orbital sigma or pi? Bonding or antibonding? (circle your answers). The orbital is between two carbons, the nuclei of which are roughly where the black circles are. 2. What atomic orbitals would be used to make the orbital from question 2? 3. Label the hybridization of each indicated atom in the following molecule as SP, SP³, or SP³, OH 4. Circle the shortest carbon-carbon single bond in the above molecule (Advanced question). Explain why it is the shortest in a sentence or two. 5. Draw the major resonance structures of the following molecule S H₂Narrow_forwardHow many of the following molecules will rotate plane polarized light? H CH3 CH3 Ме CH3 CH2CH3 H3C. CH2CH3 CI H;C. H. ČH3 Br ci' H H3CH2C 1 2 3 4 5arrow_forward
- Let us construct the molecular orbital diagram of ethylene (in pieces). a. First, construct the MO diagram of linear carbene (CH2). Draw pictures of all 6 orbitals b. Now bend the carbene to a bond angle of about 120°. How does this change your MO diagram? Draw pictures of all 6 orbitals. c. Now bring two of these carbene molecules together to make ethylene. Draw pictures of all 12 orbitals.arrow_forward4 of 4 7. Circle each of the molecule(s) below that have a a bond made from the overlap between an sp-hybridized orbital and an sp²-hybridized orbital. 8. Levomenol is a naturally occurring molecule with a sweet aroma that is used in many fragrances; it also has antimicrobial and anti-inflammatory properties. Identify the number of hybridized atoms in each category (sp, sp², sp³), o bonds, bonds, lone pairs, and implied hydrogen atoms. H OH Levomenol 9. Answer the questions below about the following molecules. A CI HO sp sp² sp³ B In which molecule would you expect the C-Cl bond to be stronger? In which molecule would you expect the C-Cl bond to be shorter? What is the geometry of the C atom in the C-Cl of molecule A? What is the geometry of the C atom in the C-Cl of molecule B? T lp H 10. The following molecule is bombykol (a naturally occurring pheromone formed in silk worms), assign a geometry and bond angle to each non-hydrogen atom.arrow_forwardWhen looking at the hybridized orbital diagram, we can see which orbitals( and their electrons) will lead to sigma, pi and lone pair of electrons. Indicate where you would find each of the following :arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning



