
Organic Chemistry (9th Edition)
9th Edition
ISBN: 9780321971371
Author: Leroy G. Wade, Jan W. Simek
Publisher: PEARSON
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 1, Problem 1.53SP
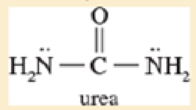
In most
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
For the following descriptions of molecules, draw the Lewis structure (showing all
atoms, lone pairs, formal charges) of the molecule and show all bond angles
(assuming ideal VSEPR angles).
An organic compound with the molecular formula H3CNO2. C is sp2
hybridized, N is sp3 hybridized, one O is sp² hybridized, while the second O is sp3
hybridized.
An organic compound with the molecular formula H2CNO*. Both C
and O are sp hybridized while N is sp³ hybridized.
A student investigates the physical and chemical properties of various carbon-containing compounds. The complete Lewis electron-dot diagrams and boiling points for two compounds, Q and X are shown in the following table.
Identify the hybridization of the valence orbitals of the carbon atom in compound X that is indicated by the arrow in the diagram.
The C-H bonds in compound Q are shorter than the C-C bonds in compound X. Explain the reason for this difference using principles of atomic structure.
For each compound, list all intermolecular forces present.
Q =
X =
Determine the molecular geometry and polarity of hydroxylamine (NH2OH), formaldehyde (CH2O), and formaldoxime (H2CNOH). Justify your deductions. Is there a difference between reactants and products for both features?.
Chapter 1 Solutions
Organic Chemistry (9th Edition)
Ch. 1.2C - a. Nitrogen has relatively stable isotopes...Ch. 1.4 - Draw Lewis structures for the following compounds....Ch. 1.5 - Write Lewis structures for the following molecular...Ch. 1.5 - Circle any lone pairs (pairs of nonbonding...Ch. 1.6 - Use electronegativities to predict the direction...Ch. 1.8 - Prob. 1.6PCh. 1.9B - Draw the important resonance forms for the...Ch. 1.9B - Prob. 1.8PCh. 1.9B - Prob. 1.9PCh. 1.9B - Use resonance structures to identify the areas of...
Ch. 1.10A - Draw complete Lewis structures for the following...Ch. 1.10B - Give Lewis structures corresponding to the...Ch. 1.10B - Prob. 1.13PCh. 1.11 - Compute the empirical and molecular formulas for...Ch. 1.16 - a. Use your molecular models to make ethane, and...Ch. 1.17 - a. Predict the hybridization of the oxygen atom in...Ch. 1.17 - Predict the hybridization geometry and bond angles...Ch. 1.17 - Predict the hybridization, geometry, and bond...Ch. 1.17 - Prob. 1.19PCh. 1.17 - Allene, CH2=C=CH2, has the structure shown below...Ch. 1.17 - 1. Draw the important resonance forms for each...Ch. 1.18B - Prob. 1.22PCh. 1.18B - Two compounds with the formula CH3CH=NCH3 are...Ch. 1.19B - Prob. 1.24PCh. 1.19B - Give the relationship between the following pairs...Ch. 1 - a. Draw the resonance forms for SO2 (bonded OSO)....Ch. 1 - Name the element that corresponds to each...Ch. 1 - Prob. 1.28SPCh. 1 - For each compound, state whether its bonding is...Ch. 1 - a. Both PCl3 and PCl5 are stable compounds Draw...Ch. 1 - Draw a Lewis structure for each species a. N2H4 b....Ch. 1 - Prob. 1.32SPCh. 1 - Prob. 1.33SPCh. 1 - Draw Lewis structures for a. two compounds of...Ch. 1 - Prob. 1.35SPCh. 1 - Some of the following molecular formulas...Ch. 1 - Prob. 1.37SPCh. 1 - Give the molecular formula of each compound shown...Ch. 1 - 1. From what you remember of electronegativities,...Ch. 1 - For each of the following structures, 1. Draw a...Ch. 1 - Prob. 1.41SPCh. 1 - Prob. 1.42SPCh. 1 - Prob. 1.43SPCh. 1 - Prob. 1.44SPCh. 1 - For each pair of ions, determine which on is more...Ch. 1 - Use resonance structures to identify the areas of...Ch. 1 - Prob. 1.47SPCh. 1 - In 1934, Edward A. Doisy of Washington University...Ch. 1 - If the carbon atom in CH2Cl2 were fat. there would...Ch. 1 - Cyclopropane (C3H6, a three-membered ring) is more...Ch. 1 - Prob. 1.51SPCh. 1 - Prob. 1.52SPCh. 1 - In most amines, the nitrogen atom is sp3...Ch. 1 - Predict the hybridization and geometry of the...Ch. 1 - Draw orbital pictures of the pi bonding in the...Ch. 1 - Prob. 1.56SPCh. 1 - Prob. 1.57SPCh. 1 - Which of the following compounds show cis-trans...Ch. 1 - Give the relationships between the following pairs...Ch. 1 - Dimethyl sulfoxide (DMSO) has been used as an...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- A particular allotrope of nitrogen, N4, was first detected in 2002. (Allotrope just means a chemical form in which an element can exist. Oxygen, for example, has two allotropes on earth: O2 and O3.) The all-nitrogen compound only lasts a short while before breaking down to N2. The compound cannot be isolated, so its structure is not yet confirmed experimentally; we can only hypothesize what its bonding and shape are. This allotrope is generally believed to be either a single ring of four nitrogens or a single chain of four nitrogens. 1. Draw the Lewis structure for both the linear form and the single ring form.arrow_forwardA particular allotrope of nitrogen, N4, was first detected in 2002. (Allotrope just means a chemical form in which an element can exist. Oxygen, for example, has two allotropes on earth: O2 and O3.) The all-nitrogen compound only lasts a short while before breaking down to N2. The compound cannot be isolated, so its structure is not yet confirmed experimentally; we can only hypothesize what its bonding and shape are. This allotrope is generally believed to be either a single ring of four nitrogens or a single chain of four nitrogens. 2. Assign formal charges to each atom in both structures. Which structure is more likely someday to be confirmed experimentally?arrow_forwardYeni Metin Belgesi - Not Defteri Dosya Düzen Biçim Görünüm Yardım For acetaldehyde (CH3CHO) write the Lewis structure. Write the hybridization and geometry of each carbon. Draw its 3D structure and write the bond angles (approximately).arrow_forward
- Draw the Lewis structures and predict the hybridization (sp, sp2, sp3), geometry (linear, trigonal, tetrahedral), and bond angles (180°, 120°, 109.5°) for the central atoms in the following compounds. C and N in C CH3CNarrow_forwardIndicate the hybridization of each of the carbon atoms in the structure below.arrow_forwardLinoleic acid is an essential fatty acid found in many vegetable oils, such as soy, peanut, and cottonseed. A key structural feature of the molecule is the cis orientation around its two double bonds, where R1 and R2 represent two different groups that form the rest of the molecule. (a) How many different compounds are possible, changing only the cis-trans arrangements around these two double bonds? (b) How many are possible for a similar compound with three double bonds?arrow_forward
- Draw the Lewis diagram of Serotonin. Mark the electron geometry and molecular geometry of 6 interior atoms. Draw the Serotonin structure using skeletal notation here. Mark all sigma bonds, all pi bonds, and the hybridization of all interior atoms.arrow_forwardMethyl isocyanate, CH3 -N= C = O, is used in the industrial synthesis of a type of pesticide and herbicide known as a carbamate. As a historical note, an industrial accident in Bhopal, India, in 1984 resulted in leakage of an unknown quantity of this chemical into the air. An estimated 200,000 people were exposed to its vapors, and over 2000 of these people died. Q.) Write a Lewis structure for methyl isocyanate and predict its bond angles. What is the hybridization of its carbonyl carbon? Of its nitrogen atom?arrow_forwardChloral, Cl₃C-CH=O, reacts with water to form the seda-tive and hypnotic agent chloral hydrate, Cl₃C-CH(OH)₂. DrawLewis structures for these substances, and describe the change inmolecular shape, if any, that occurs around each of the carbonatoms during the reactionarrow_forward
- Acrylic fibers are polymers made from a starting material called acrylonitrile, H2C(CH)CN. In acrylonitrile, a - CN group replaces a hydrogen atom on ethene. Draw the Lewis diagram for this molecule, give the hybridization of each carbon atom, and describe the \pi orbitals and the number of electrons that occupy each one. Draw the three-dimensional structure of the molecule, showing all angles.arrow_forwardsuppose you forget to take into account the presence of unshared pair of electrons on nitrogen in the molecule nh3. what would you then predict for the h-n-h bond angles and geometry of ammonia?arrow_forwardTwo useful organic compounds that contain Cl atoms are vinyl chloride (CH2 = CHCI) and chloroethane (CH;CH;CI). Vinyl chloride is the starting material used to prepare poly (vinyl chloride), a plastic used in insulation, pipes, and bottles. Chloroethane (ethyl chloride) is a local anesthetic. Why is the C – Cl bond of | vinyl chloride stronger than the C – Cl bond in chloroethane.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
INTRODUCTION TO MOLECULAR QUANTUM MECHANICS -Valence bond theory - 1; Author: AGK Chemistry;https://www.youtube.com/watch?v=U8kPBPqDIwM;License: Standard YouTube License, CC-BY