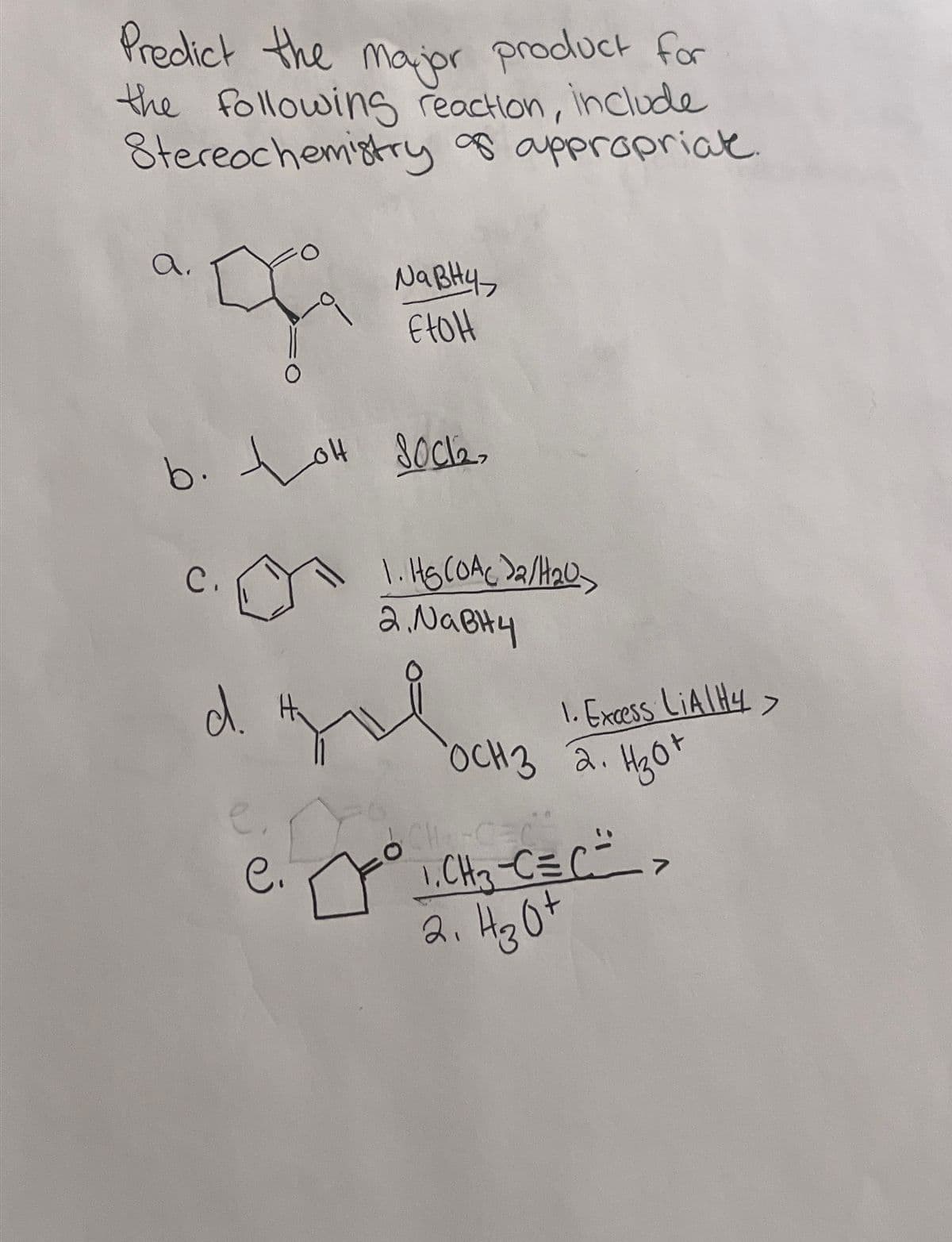
Predict the major product for the following reaction, include Stereochemistry of appropriat a. NaBH4 EtOH b. o socke to C. 1. HS (OAc)2/H20 2. NaBH4 d. 1. Excess LiAlH4> OCH 3 2. H3O+ CH-C e. C. ° 1. CH₂-C=C => 2. H30+
Q: B. Carbon monoxide, CO, is poisonous because it bonds more tightly to the hemoglobin in red blood…
A: Carbon monoxide is poisonous because it bonds more tightly to the hemoglobin in red blood cells than…
Q: Draw Zaitsev and Hofmann products that are expected when each of the following compounds is treated…
A: An arrow always depicts from a region of high electron density to low electron density ; that is…
Q: What will happen in column chromatography if MPB purification buffer was used instead of typical…
A: Column chromatography is used for separating and purifying compounds from a mixture.Column…
Q: Ti6Al4V is a high-strength alloy of titanium widely used in industry. Show work..don't give…
A: In the given question we have to calculate the mass of titanium obtained from the given mole of…
Q: Rank the following carboxylic acid derivatives in decreasing order (most to least) of reactivity…
A:
Q: Draw the major organic product of the reaction below. H3C H KOH, H₂O CH3
A:
Q: Use the Henderson-Hasselbalch equation to calculate the pH of each solution: a solution that is 0.28…
A: The objective of this question is to calculate the pH of a solution using the Henderson-Hasselbalch…
Q: 4. Propose reagents for the synthesis of the target carboxylic acid from each of the indicated…
A: The objective of the question is to propose reagents for the synthesis of a carboxylic acid from the…
Q: For each pair of compounds below, identify the more acidic compound. × Your answer is incorrect. H…
A: In this question, we have to find out which ion or molecule is the stronger acid :HCl or H3O+
Q: What is the original molarity of a solution of ammonia whose pH is 10.54? Be sure your answer has…
A:
Q: Hydrogen produced from a hydrolysis reaction was collected over water. The data is compiled in the…
A:
Q: At a certain temperature the value of the equilibrium constant, Kc, is 0.8375 for the reaction. SiO2…
A: • Equilibrium constant for reverse reaction:For reverse reaction the value of the equilibrium…
Q: Predict the major product for the following reaction. CN 1) xs LiAlH4 2) H₂O ? Modify the given…
A: The major organic product of the reaction is 2-methylbutan-1-amine. Explanation:
Q: A chemist is studying the following equilibirum, which has the given equilibrium constant at a…
A: The equilibrium pressure of = ?
Q: An analytical chemist is titrating 109.4 mL of a 0.7700 M solution of hydrazoic acid (HN3) with a…
A: We will first calculate the millimole of both acid and base. Then we will find whether weak acid or…
Q: Draw the appropriate Newman projection which leads to the major E2 ELIMINATION product. Hint 1:…
A: For the E2 elimination reaction to take place the leaving group and hydrogen should be anti-peri…
Q: NH3 H H
A:
Q: Consider the following chemical equilibrium: HCOO- (aq) + H2O(aq) = HCOOH (aq) + OH- (aq). Which of…
A: option c.(III) Explanation:Step 1: The addition of HCl to the solution will cause decrease in pH ,…
Q: H₂C CH₂ NH3 slightly acidic (pH 4-5) 1) LIAIH in diethyl ether 2) H₂O
A: The carbonyl compounds have an electrophilic C and ammonia has nucleophilic N atom. So, the…
Q: Question 5 Draw Zaitsev and Hofmann products that are expected when each of the following compounds…
A:
Q: 1. Li(CH3)2Cu 1. CH3MgBr / dry ether a. d. H₂NNH2/KOH g. j. (C6H5)3P+-CH2 2. H3O+ 2. H3O+ 1. NaBH4…
A: We have to synthesize the given compounds from the starting material 2-cyclohexenone.
Q: Which of the following orbitals contains fewer electrons in a ruthenium(II) cation than a neutral…
A: The objective of this question is to determine which orbital in a ruthenium(II) cation contains…
Q: Draw the starting material that, under the given reaction conditions, results in the following…
A: The objective of this question is to draw the starting compounds in the given reactions.
Q: 2. IN HNO3 H2SO4 % o/p/m?
A: Information about the question
Q: Consider the following reaction where Kc = 55.6 at 698 K.H2(g) + I2(g) 2HI(g)A reaction mixture was…
A: The objective of the question is to determine whether the given reaction is at equilibrium or not.…
Q: Use the Henderson-Hasselbalch equation to calculate the pH of each solution: a solution that is 0.28…
A: The objective of the question is to calculate the pH of two different solutions using the…
Q: what is the name of the structure OH о N
A: IUPAC is an acronym for the International Union of Pure and Applied Chemistry. It is an…
Q: Which statements are true about weak acids? They dissociate completely in water. The Ka values are…
A: Statements about weak acid.
Q: Consider the following three compounds. Part: 0/2 Part 1 of 2 Rank the three compounds in order of…
A: We need to determine the order of heat of hydrogenation for given alkenes.Organic compounds that…
Q: 2. Fill in the boxes in the following problem. (Hints: The difference between the # of carbons in…
A: The missing reactant, reagents and product has find with proper explanation. Also we have provided…
Q: Hypoiodous acid (HIO) is used as a strong oxidizing agent. The K₂ of HIO is 2.3x10-11 at 25°C. What…
A: Hypoiodous acid (HIO) = 0.874 MKa of HIO is 2.3×10-11 at 25°C. Calculate the pOH of hypoiodous acid.
Q: Which option contains the most acidic proton? ६ ६ ४ ६ a. b. C. d.
A: >A-H ---> A- + H+Greater the stability of conjugated base (A-) stronger is the conjugate…
Q: 5. Give number of valence electrons for the following compounds and account for their…
A: Since you have posted a question with multiple sub-parts, we will provide the solution only for the…
Q: Provide an acceptable name for the following compound. 0- CH-N-CH2CH3 Spell out the full name of the…
A: RULES for IUPAC nomenclature; In first step, we identify functional groups present. We make longest…
Q: Br H Cl H Enter the correct IUPAC nomenclature. cis-1-bromo
A: For naming a compound according to IUPAC, there are some rules. They are -Find the longest chain or…
Q: The following H-NMR spectrum (showing all available signals) is for one of the possible products…
A: A triplet at 1.1 ppm and a quartet at 2.4 ppm suggests that the molecule has -CH2CH3 group. The…
Q: HO для aqueous H₂SO + NaCN •You do not have to consider stereochemistry.
A: Given reactionWe have to find the product.Note:Do not have to consider stereochemistry
Q: Question 13 Draw major E2 ELIMINATION product. Hint: Review Chapter 10.3, the elimination can only…
A: In this reaction E2 reaction has been done and the final product is an alkene.
Q: 35.5 g O, in a 15.7 L container at 1.25 atm ☐ K x10
A: Given that,Mass of Molar mass of Volume of the gas= Pressure of the gas in the container=…
Q: 1. Write a mechanism for the formation of oximes from aldehydes and hydroxylamine. 2. Reaction of…
A: The objective of the first part of the question is to describe the mechanism for the formation of…
Q: One cup of fresh orange juice contains 139 mg of ascorbic acid (vitamin C, C6H8O6). Part A Given…
A: Given:Mass of ascorbic acid in one cup of fresh orange juice = 139 mg = 0.139 g Volume of one cup =…
Q: In radical chlorination of alkanes, non-equivalent hydrogens react with chlorine atoms at different…
A: Relative reactive rates of primary secondary and tertiary C-H bonds = 1 : 3.9 : 5.2Temperature = For…
Q: 9.49 Draw a stepwise mechanism for the following reaction.
A: In an E2 reaction, the abstraction of the acidic proton by the base and the loss of the leaving…
Q: 100.0-mL buffer solution is 0.175 M in HClO and 0.150 M in NaClO. What is the initial pH of this…
A: The volume of buffer solution = The concentration of The concentration of The mass of The mass of
Q: If 36.7 g of AgNO₃ react with 28.6 g of H₂SO₄ according to this UNBALANCED equation below, how many…
A: The objective of this question is to determine the amount of Ag2SO4 that could be formed from the…
Q: Can someone write the IUPAC names for these compounds?
A: RULES for IUPAC nomenclature; In first step, we identify functional groups present. We make longest…
Q: Identify the Major and ALL Minor product(s) that are expected for each of the following reactions.…
A: Nucleophile: NaSMeLeaving group: OMe
Q: Draw the major product of this reaction. Use a dash or wedge bond to indicate stereochemistry of…
A: In this reaction alkyl bromide has been turned into amine.
Q: Identify the functional groups in the following molecules. (Use names from the table below. List…
A: In the given question, two molecules are given. We have to find out the functional groups present in…
Q: Calculating the pH of a weak acid titrated with a strong base 0/5 Izabella An analytical chemist is…
A: Ph of the solution is 3.82Explanation:To calculate the pH of the solution after adding the NaOH…
Unlock instant AI solutions
Tap the button
to generate a solution
Click the button to generate
a solution
- when H2O2 is added into acidified K2Cr2O7 which colour appears?In light of your answer to Problem 30-40, explain why a mixture of products occurs in the following reaction:Phenols (ArOH) are relatively acidic, and the presence of a substituent group on the aromatic ring has a large effect. The pKa of unsubstituted phenol, for example, is 9.89, while that of p-nitrophenol is 7.15. Draw resonance structures of the corresponding phenoxide anions and explain the data.
- Kindly share detailed explanation for each step. All related reactions of k4fecn6 and k3fecn6 with fe 2+ and fe3+ may be shared.What is (are) the elimination product(s) obtained from the following reaction? A)only I B)I and II C) I,II, and ,III D) II, III, and IVReally hoping for solutions since I’m having a hard time with this. Pls. skip if unsure or not willing to answer the subitems (these are all connected for one item). Thanks in advanced. A kinetics experiment was performed for the formation of a novel compound, UNO, from its reactants, Chemicals G, R, A, D, and E. The first six runs (Runs 1-6) were all performed at 301 K, while the next two runs (Runs 7-8) at varying temperatures. These are tabulated in the table shown below. Table 1. Reaction rate for the reaction of the different chemicals at a given concentration at 301 K. Run [G], M [R], M [A], M [D], M [E], M Rate, M/s 1 0.80 0.01 0.20 0.50 2.00 0.180 2 0.80 0.02 0.20 0.50 0.02 3.54 x 10-3 3 0.80 0.01 0.20 0.50 0.02 1.80 x 10-3 4 0.80 0.01 0.60 0.50 0.02 1.80 x 10-3 5 1.20 0.01 0.20 0.50 0.02 4.02 x 10-3 6 0.80 0.01 0.20 0.15 0.02 1.95 x 10-2 a. Calculate the rate order of each chemical and overall rate order of the…
- Balence the following half reation under basic conditions: PbO2 → Pb2-What would the final products look like? Pls specify stereochemistry if neededWhich reagent(s) sequence(s) is/are optimum for preparing butanoic acid from 1-propanol? i. K2Cr2O7 in acidii. (1) PBr3; (2) NaCN; (3) H2O; (4) [H+] iii. (1) NaCN; (2) H2O; (3) [H+]iv. LiAlH4
- 2A---> 2B + C (Kp1) 2D---> 2E + C (Kp2) Now find the corresponding Kp3= f(Kp1, Kp2) = ?for this reaction: D + B----> A + E elaborate throughly with every step and annotationPredict the product of the following reaction and classify the reaction Pb+FeSO4---> PbSO4+ ______Why does the final product has the opposite configuration compared to the reactant? Shouldn’t it form OMs first, then OMs gets substituted by Cl- via Sn2 (the 1st inversion of configuration) then the Cl- gets substituted by OCH3- (the 2nd inversion of configuration? To my understanding 2 inversions = same configuration



