
Label each ether and alcohol in brevenal, a marine natural product. Classify each alcohol as
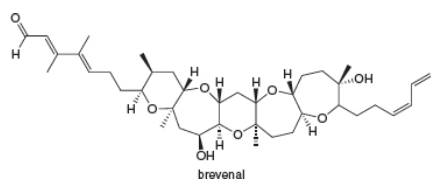
Interpretation: Each ether and alcohol in brevenal is to be labeled and each alcohol is to be classified as
Concept introduction: Alcohols and ethers are the functional groups that involve
Answer to Problem 9.1P
Each ether and alcohol in brevenal is labeled as shown below.
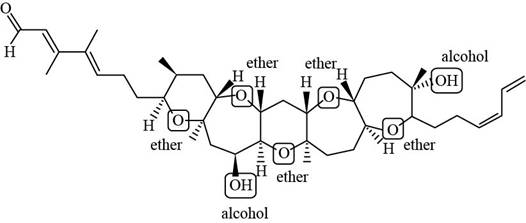
Explanation of Solution
The structure of brevenal (given compound) is,
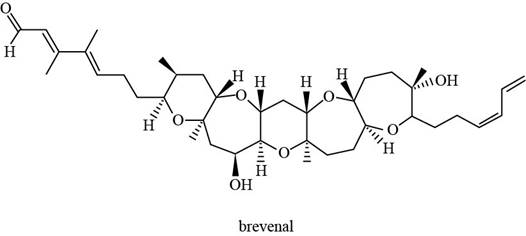
Figure 1
Alcohols and ethers are the functional groups that involve
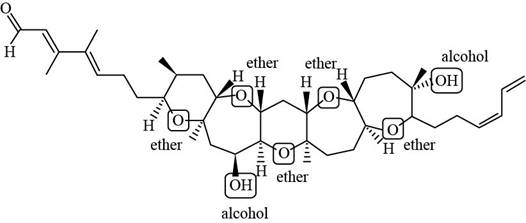
Figure 2
Alcohols are classified as
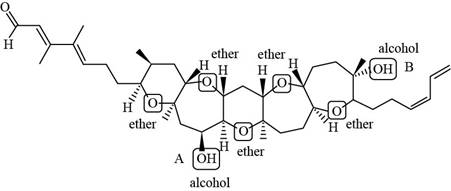
Figure 3
In Figure 3, the carbon atom of alcohol (A) is bonded to two
If the number of carbon atom bonded to the carbon atom with the hydroxyl group is one, then it is classified as
Thus, each alcohol in brevenal is classified as,
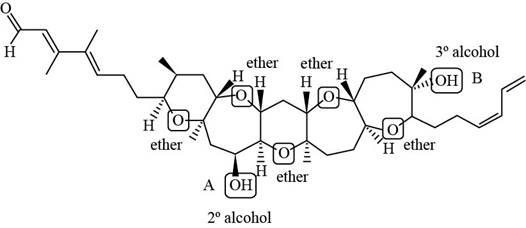
Figure 4
Each ether and alcohol in brevenal is labeled in Figure 2.
Want to see more full solutions like this?
Chapter 9 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Chemistry & Chemical Reactivity
Organic Chemistry - Standalone book
Chemistry
Chemistry: Matter and Change
Principles of Chemistry: A Molecular Approach (3rd Edition)
- Draw and name ethers and heterocyclic ethers, including epoxides. Predict theirrelative boiling points, solubilities, and solvent properties.arrow_forwardTHC is the active component in marijuana, and ethanol is the alcohol in alcoholic beverages. Explain why drug screenings are able to detect the presence of THC but not ethanol weeks after these substances have been introduced into the body. CH3 OH CH;CH2-OH ethanol CH3 ČH3 (CH),CH3 tetrahydrocannabinol THCarrow_forwardClassify each alkyl halide as 1 °, 2 °, or 3 °.arrow_forward
- 1: Give at least five (5) uses of Alcohol and Phenol Name Functional Group R-OH Alcohols LOH Phenols Based on the illustration above. What is the difference between alcohol and phenol? Can Phenol react with alcohol?arrow_forwardOrganic Reaction Write an equation for the oxidation of each alcohol. Use [O] above the arrow to indicate an oxidizing agent. If no reaction occurs, write "no reaction" after the arrow. 1. CH3CH2CH2CH2CH2OH CH₂ CH₂CCH₂CH₂ ОН CH CHCH CH CH CH, ОНarrow_forwardPredict which member of each group is most soluble in water, and explain the reasons for your predictions. phenol, cyclohexanol, or 4-methylcyclohexanolarrow_forward
- Give the IUPAC name for each alcohol но сн CH,CHCCH,CH,CH3 (CH)½CHCH,CHCH,CH3 b. a. CH,CH,CH,OH HO. с. CH,CH,CH,CH, Draw the products formed when each alcohol is dehydrated with H2SO4. Use the Zaitsev rule to predict the major product when a mixture forms. OH он b. -CHCH,CH3 а. Он с. CH3CHCH2CH,CH;CH3arrow_forwardLabel each functional group as an alcohol, ether, acetal, or hemiacetal. LOCH3 а. OCH3 (select) ОН b. CH3-C-OCH,CH2CH3 CH3 (select) с. OCH,CH,CH3 alcohol and ether v OCH,CH,CH3 (select)arrow_forwardName each of the following alcohols. HO CHI CHO он OH H2d-CH-CH3 НЗС H3C-CH2-CH2-CH-CH2-CH2-OH H2C-CH3 왜 I H3C-CH2-C-CH2-CH3 CH3arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





