
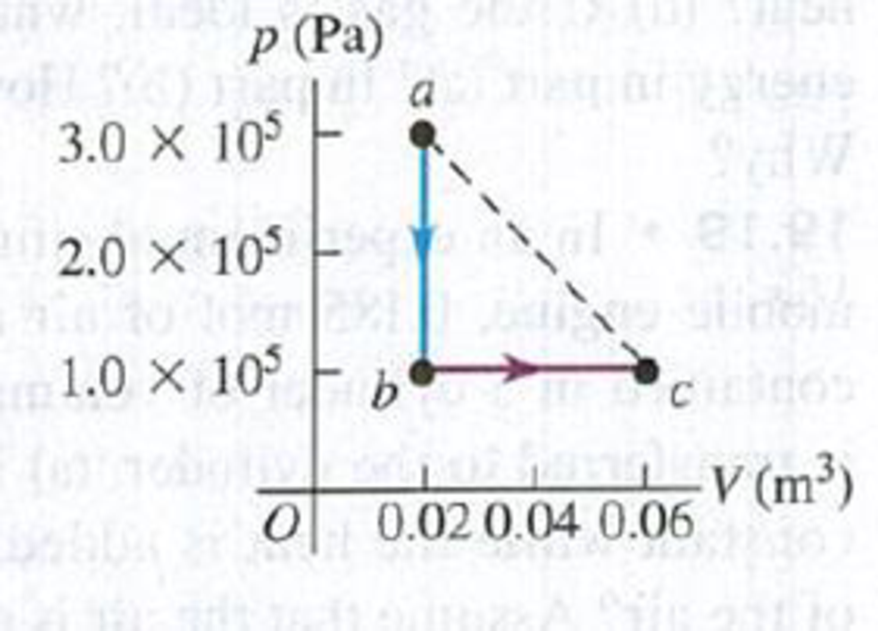
A volume of air (assumed to be an ideal gas) is first cooled without changing its volume and then expanded without changing its pressure, as shown by path abc in Fig. P19.39. (a) How does the final temperature of the gas compare with its initial temperature? (b) How much heat does the air exchange with its surroundings during process abc? Does the air absorb heat or release heat during this process? Explain. (c) If the air instead expands from state a to state c by the straight-line path shown, how much heat does it exchange with its surroundings?
Figure P19.39
Want to see the full answer?
Check out a sample textbook solution
Chapter 19 Solutions
University Physics (14th Edition)
Additional Science Textbook Solutions
Conceptual Physics (12th Edition)
College Physics (10th Edition)
Cosmic Perspective Fundamentals
Essential University Physics: Volume 1 (3rd Edition)
College Physics: A Strategic Approach (3rd Edition)
Tutorials in Introductory Physics
- In Figure P19.22, the change in internal energy of a gas that is taken from A to C along the blue path is +800 J. The work done on the gas along the red path ABC is 500 J. (a) How much energy must be added to the system by heat as it goes from A through B to C? (b) If the pressure at point A is five times that of point C, what is the work done on the system in going from C to D? Figure P19.22 (c) What is the energy exchanged with the surroundings by heat as the gas goes from C to A along the green path? (d) If the change in internal energy in going from point D to point A is +500 J, how much energy must be added to the system by heat as it goes from point C to point D?arrow_forwardFigure P21.45 shows a cyclic process ABCDA for 1.00 mol of an ideal gas. The gas is initially at Pi = 1.50 105 Pa, Vi = 1.00 103 m3 (point A in Fig. P21.45). a. What is the net work done on the gas during the cycle? b. What is the net amount of energy added by heat to this gas during the cycle? FIGURE P21.45arrow_forwardA cylinder is closed at both ends and has insulating EZZ3 walls. It is divided into two compartments by an insulating piston that is perpendicular to the axis of the cylinder as shown in Figure P21.75a. Each compartment contains 1.00 mol of oxygen that behaves as an ideal gas with = 1.40. Initially, the two compartments haw equal volumes and their temperatures are 550 K and 250 K. The piston is then allowed to move slowly parallel to the axis of the cylinder until it comes to rest at an equilibrium position (Fig. P2l.75b). Find the final temperatures in the two compartments.arrow_forward
- In Figure P17.32, the change in internal energy of a gas that is taken from A to C along the blue path is +800 J. The work done on the gas along the red path ABC is 500 J. (a) How much energy must be added to the system by heat as it goes from A through B to C? (b) If the pressure at point A is five times that of point C, what is the work done on the system in going from C to D? (c) What is the energy exchanged with the surroundings by heat as the gas goes from C to A along the green path? (d) If the change in internal energy in going from point D to point A is +500 J, how much energy must be added to the system by heat as it goes from point C to point D? Figure P17.32arrow_forwardA 2.00-mol sample of a diatomic ideal gas expands slowly and adiabatically from a pressure of 5.00 atm and a volume of 12.0 L to a final volume of 30.0 L. (a) What is the final pressure of the gas? (b) What are the initial and final temperatures? Find (c) Q, (d) Eint, and (e) W for the gas during this process.arrow_forward(a) Determine the work done on a gas that expands from i to f as indicated in Figure P19.16. (b) What If? How much work is done on the gas if it is compressed from f to i along the same path? Figure P19.16arrow_forward
- A gas in a cylindrical closed container is adiabatically and quasi-statically expanded from a state A (3 MPa, 2 L) to a state B with volume of 6 L along the path 1.8pV= constant. (a) Plot the path in the pV plane. (b) Find the amount of work done by the gas and the change in the internal energy of the gas during the process.arrow_forwardA certain ideal gas has a molar specific heat of Cv = 72R. A 2.00-mol sample of the gas always starts at pressure 1.00 105 Pa and temperature 300 K. For each of the following processes, determine (a) the final pressure, (b) the final volume, (c) the final temperature, (d) the change in internal energy of the gas, (e) the energy added to the gas by heat, and (f) the work done on the gas. (i) The gas is heated at constant pressure to 400 K. (ii) The gas is heated at constant volume to 400 K. (iii) The gas is compressed at constant temperature to 1.20 105 Pa. (iv) The gas is compressed adiabatically to 1.20 105 Pa.arrow_forward
 Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning
Physics for Scientists and Engineers: Foundations...PhysicsISBN:9781133939146Author:Katz, Debora M.Publisher:Cengage Learning Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers, Technology ...PhysicsISBN:9781305116399Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
 Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and EngineersPhysicsISBN:9781337553278Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Physics for Scientists and Engineers with Modern ...PhysicsISBN:9781337553292Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning





