Organic Chemistry: A Guided Inquiry
2nd Edition
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Andrei Straumanis
Chapter4: Polar Bonds, Polar Reactions
Section: Chapter Questions
Problem 4E
Related questions
Question

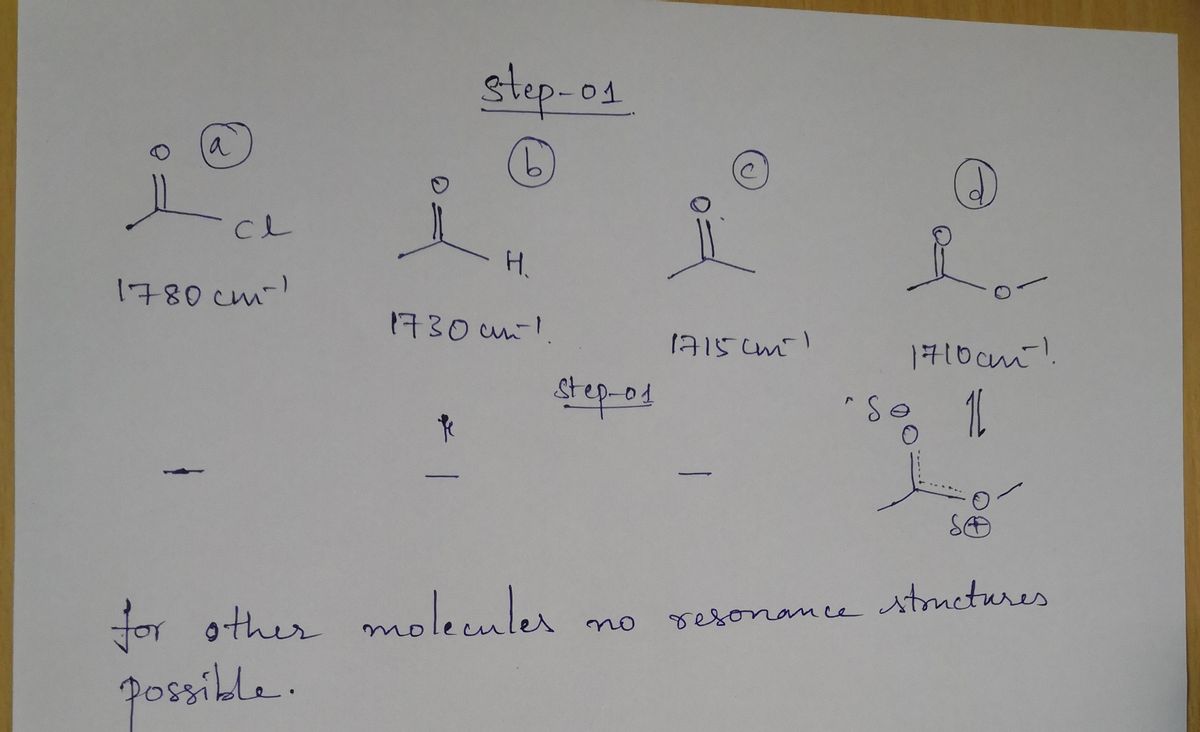
Transcribed Image Text:4 The carbonyl functional groep C =0 tends to
cm. In finth, there is
show
ardund 1700
Some
variability in exactly where the C =0 stretch
is are
Data is,
awith shorter bonds. Some are at sematter values,
consistent with longer bonds.
D
e
CI
17 80cm
H
1730cm
1710 cm
• Provide the next-best resonemce structure for four
of
CH3
1715cm
• Pick
any
two and state how the resonance structures
can be word to help correlate structures with the ghen values
for stething frequencies.
Expert Solution
Step 1
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:
9780618974122
Author:
Andrei Straumanis
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,