A chemist is studying the following equilibirum, which has the given equilibrium constant at a certain temperature: 2 NO(g) + Cl₂(g) = 2 NOCI (g) K₂=1.x 10-5 He fills reaction vessel at this temperature with 5.0 atm of nitrogen monoxide gas and 6.7 atm of chlorine gas. Use this data to answer the questions in the table below. n yes Can you predict the equilibrium pressure of NOCI, using only the tools available to you within ALEKS? X 3 ? If you said yes, then enter the equilibrium pressure of NOCI right. Round your answer to 1 significant digit. o no Da atm
A chemist is studying the following equilibirum, which has the given equilibrium constant at a certain temperature: 2 NO(g) + Cl₂(g) = 2 NOCI (g) K₂=1.x 10-5 He fills reaction vessel at this temperature with 5.0 atm of nitrogen monoxide gas and 6.7 atm of chlorine gas. Use this data to answer the questions in the table below. n yes Can you predict the equilibrium pressure of NOCI, using only the tools available to you within ALEKS? X 3 ? If you said yes, then enter the equilibrium pressure of NOCI right. Round your answer to 1 significant digit. o no Da atm
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter17: Equilibrium
Section: Chapter Questions
Problem 1ALQ: Consider an equilibrium mixture of four chemicals (A. B. C. and D. all gases) reacting in a closed...
Related questions
Question
What's the answer to this?

Transcribed Image Text:O KINETICS AND EQUILIBRIUM
=
Valentina V
Using the small x approximation to solve equilibrium problems
2/5
A chemist is studying the following equilibirum, which has the given equilibrium constant at a certain temperature:
-5
2 NO(g) + Cl₂(g) = 2 NOC1 (g)
K₂= 1. x 10
P
He fills a reaction vessel at this temperature with 5.0 atm of nitrogen monoxide gas and 6.7 atm of chlorine gas. Use this data to answer the questions in the
table below.
yes
x10
Can you predict the equilibrium pressure of NOC1, using only the
tools available to you within ALEKS?
no
X
Ś
?
If you said yes, then enter the equilibrium pressure of NOCI at
right. Round your answer to 1 significant digit.
atm
Explanation
Check
© 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility
A
000
Ar
B
KI
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

Follow-up Questions
Read through expert solutions to related follow-up questions below.
Follow-up Question
I have a few more, heres one
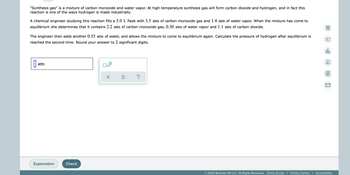
Transcribed Image Text:"Synthesis gas" is a mixture of carbon monoxide and water vapor. At high temperature synthesis gas will form carbon dioxide and hydrogen, and in fact this
reaction is one of the ways hydrogen is made industrially.
A chemical engineer studying this reaction fills a 5.0 L flask with 3.3 atm of carbon monoxide gas and 1.4 atm of water vapor. When the mixture has come to
equilibrium she determines that it contains 2.2 atm of carbon monoxide gas, 0.30 atm of water vapor and 1.1 atm of carbon dioxide.
The engineer then adds another 0.35 atm of water, and allows the mixture to come to equilibrium again. Calculate the pressure of hydrogen after equilibrium is
reached the second time. Round your answer to 2 significant digits.
atm
Dx1
x10
X
5
?
Explanation
© 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility
Check
A
000
Ar
81
K
Solution
Follow-up Question
This is my last one, thanks!
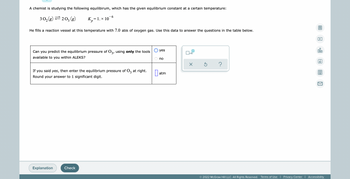
Transcribed Image Text:A chemist is studying the following equilibirum, which has the given equilibrium constant at a certain temperature:
6
30₂(g)=203 (g)
= 1. × 10
He fills a reaction vessel at this temperature with 7.0 atm of oxygen gas. Use this data to answer the questions in the table below.
▷
yes
000
x10
Can you predict the equilibrium pressure of 03, using only the tools
available to you within ALEKS?
no
Ar
X
Ś ?
If you said yes, then enter the equilibrium pressure of O3 at right.
Round your answer to 1 significant digit.
atm
Explanation
Check
© 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility
Bp
0:
Solution
Follow-up Question
this is my next question
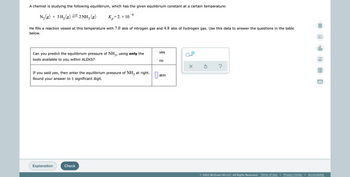
Transcribed Image Text:A chemist is studying the following equilibirum, which has the given equilibrium constant at a certain temperature:
6
N₂(g) + 3 H₂(g) ⇒ 2NH3 (g)
K₂=2. × 10
р
He fills a reaction vessel at this temperature with 7.0 atm of nitrogen gas and 4.8 atm of hydrogen gas. Use this data to answer the questions in the table
below.
yes
Can you predict the equilibrium pressure of NH3, using only the
tools available to you within ALEKS?
x10
no
X
Ś
?
If you said yes, then enter the equilibrium pressure of NH3 at right.
Round your answer to 1 significant digit.
atm
Explanation
Check
© 2022 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility
0:
A
Ar
|
Solution
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Introduction to General, Organic and Biochemistry
Chemistry
ISBN:
9781285869759
Author:
Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar Torres
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning