Give the product of the reaction shown below. Would the enantiomer or a diastereomer of the molecule below yield the same product? Show the product for either case.. Ph Br NaOH Ph Δ
Give the product of the reaction shown below. Would the enantiomer or a diastereomer of the molecule below yield the same product? Show the product for either case.. Ph Br NaOH Ph Δ
Organic Chemistry
8th Edition
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Chapter9: Nucleophilic Substitution And Β-elimination
Section: Chapter Questions
Problem 9.39P: Following are diastereomers (A) and (B) of 3-bromo-3,4-dimethylhexane. On treatment with sodium...
Related questions
Question
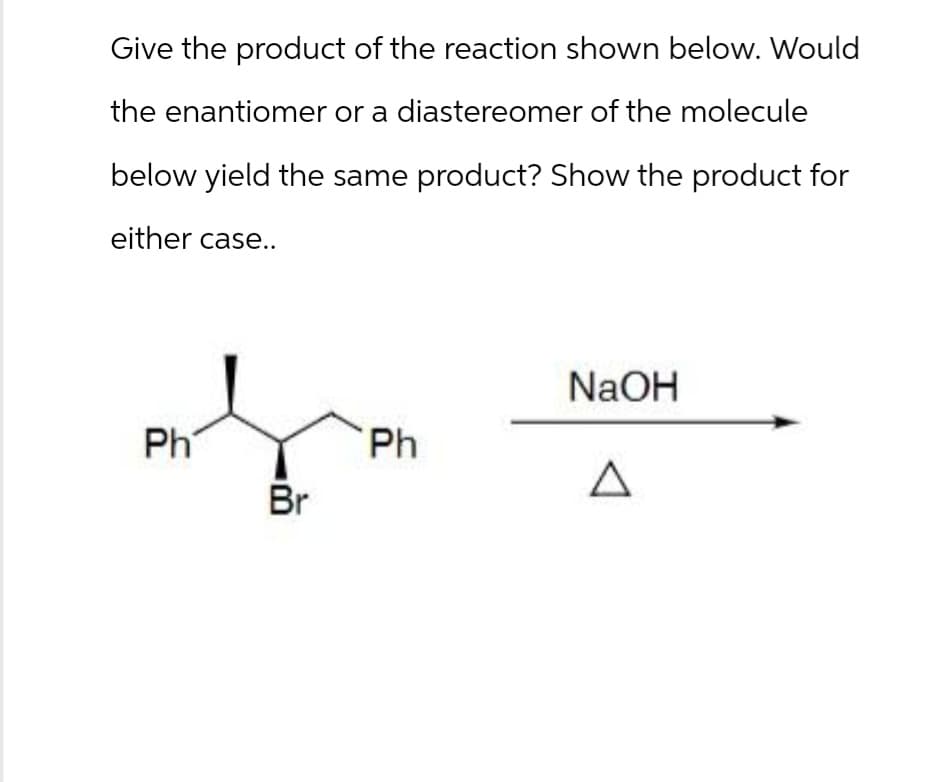
Transcribed Image Text:Give the product of the reaction shown below. Would
the enantiomer or a diastereomer of the molecule
below yield the same product? Show the product for
either case..
Ph
Br
NaOH
Ph
Δ
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

Recommended textbooks for you

Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning


Organic Chemistry
Chemistry
ISBN:
9781305580350
Author:
William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:
Cengage Learning
