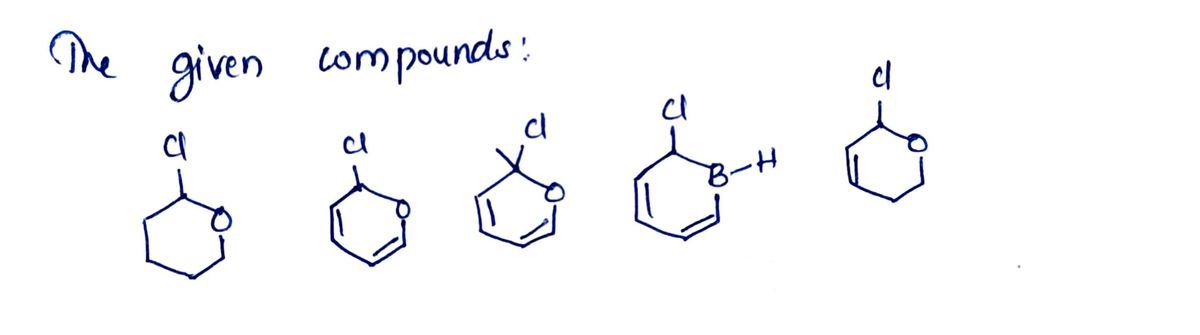
3. Rank the following in order by the rate in which they could participate in an SN1 reaction (“1" being the fastest and "5" being the slowest to react). CI CI B-
Q: sborie Il.A galvanic cell is based on the reaction priwbnbon MnO, (aq) + H*(aq) + CI0,-(aq) → CIO, (...
A:
Q: 656 486 434 410) longer the wavelength, t Hydrogen Emission Spectrum wavelength, A (nm) Figure 1. Fl...
A:
Q: A person contains 7L of blood. Blood is composed of 55 percent plasma and 45 percent blood cells by ...
A: Given, A person's body has 7L blood. Blood plasma - 55 %. Blood cells - 45 %. Density of blood plasm...
Q: Name the organic reactant required to react with ammonia to produce hexan-1-amine and HBr.
A:
Q: What is the mass in kg of 0.49 mol CO2?
A: Number of moles of CO2 = 0.49 mol Mass of CO2 (in kg) = ?
Q: Triglycerides are esters of fatty acids with ______ Fill in the blanck.
A:
Q: . A certain gas obeys the van der Waals equation with a = 0.50 m6 Pa mol-2. Its volume is found to b...
A:
Q: 1. Given the following reduction potentials: Ag*(aq) + e → Ag(s) Zn (aq) + 20ZN(s) Calculate the cel...
A: 1) Zn2+(aq) + 2Ag(s) →Zn (s) + Ag2+(aq) Oxidation reaction : 2Ag ----> 2Ag+ + 2e- (Anode) R...
Q: Determine if each of the following molecules is polar or non-polar. H HH HH H HHH HH HH H. Br H H-C-...
A: It is a non-polar molecule as there is no extra atom attached other than C and H. Here, the electron...
Q: Prepared buffer solution Given Ammonia Volume = 68 mL | Ammonia Concentration = 0.17 M Ammonium Chl...
A: Th working formula is V1×S1 = V2×S2 V1 = initial volume V2 = final volume S1 = initial concentration...
Q: 3 Draw the structural formula for; a ketone (C4H$O) an amine (C3H13N) vi. an alcohol (C2H6O) an ethe...
A: Structures of the given organic compounds: As per our guideline we have to answer first three qu...
Q: It is a general term that refers to the properties of the solutions that depend on the quantity of ...
A:
Q: a gallon of water weighs about 4.9 kg. how many gallons of water are there in 29.4kg?
A: Given, Weight of 1 gallon of water is 4.9 kg.
Q: energy (ir
A:
Q: What happens when you add Benedict's reagent to acetaldehyde? Will there be a percipitate? If yes, w...
A: Benedict reagent can be prepared by reaction of copper (II) sulfate pentahydrate with sodium citrat...
Q: A student pharmacist prepared a buffer solution by mixing 68 ml of 0.17 M ammonia and 42 mL of 0.12 ...
A:
Q: Name the major product for the reaction of aqueous bromoethane with NaSH.
A: Substitution reaction is the reaction in which one species is replaced by other.
Q: Calculate The number of atoms 0.1g of ozone
A: We know that, 1 mol = 6.023×1023 number of molecules.
Q: :0-R RーC-る R
A:
Q: The hydrolysis of solid urea is given by the reaction: H20 (I) + H2NCONH2 → CO2 (g) + 2 NH3 (g) Calc...
A:
Q: How much THE REQUIRED AMOUNT OF 500mg ASCORBIC ACID TABLETS should i need to pulverize? and; How muc...
A: Concentration of stock solution of ascorbic acid = 0.02 M Av...
Q: Answer the following questions briefly. 1. What is the use of benedicts test? 2. What are the reag...
A: This question belongs to practical organic chemistry that is structural identification of organic co...
Q: a. N2(g) + C2H2(g) – 2HCN(g) b. C3H5C2H5(g) – C3H5CH:CH2(g) + H2(8) Determine the standard heat for ...
A:
Q: How much 6.00% sucrose solution must be added to 5.00 liters of 60.0% sucrose solution to dilute it ...
A:
Q: OH SO4? PO43 Na" Al H+1
A: While writing a chemical formula from combining cation and anion, we must ensure the charge balance....
Q: :0-R R-C - ö: R
A: Based on the groups or functional groups attached, organic compounds are named differently.
Q: CH2=CH(CH2)4CH3 Give the Expanded Structure and IUPAC Name
A: The IUPAC name of the molecule is hept-1-ene.
Q: serum uric acid concentration is 0.80 mmol/L. express this in mg/dl
A: The answer, 0.8 mmol/L = 13.449 mg/dl.
Q: НО. ОН
A: Addition of hydrogen to a molecule or removal of oxygen is called reduction. Removal of hydrogen to ...
Q: | R- C-OH 36 R-C-R 37 R-C-H 38 | R - с-OR 39 | R- C- NH -R 40 O=
A: Answer Which species has some dipole moment are polar and which speci...
Q: The flying out of BB's out of the petri dish in the isotope experiment, represented O a. ice cores O...
A: Isotopes are elements which have the same number of protons as the corresponding element in the peri...
Q: 1 10 9 8 7 6 5 4 3 2 1 HSP-00-768 מממ
A: Looking at the multiplicity(splitting) of the signals, a triplet means there are two neighbouring hy...
Q: 1. outline a procedure for separating a mixture of naphthalene (a neutral compound), toluic acid (an...
A: “Since you have asked multiple question, we will solve the first question for you. If you want any s...
Q: Draw: 4-Mercapto-3-methylthiopentene
A: we can write the structure of the given compound based on the position of the different substituents...
Q: Consider the following unbalanced particulate representation of a chemical equation: O = red F = gre...
A:
Q: How many grams of CO2 are formed from the combustion of 129 g methane?
A: The above question can be solved by using stoichiometric calculations.
Q: How many grams of platinum (Pt) contain the same number of atoms as 5.0 g of lithium (Li)? О а. 0.18...
A: Given, Mass of lithium (Li) = 5.0 g Mass (in grams) of platinum (Pt) contain the same number of atom...
Q: How much diluent is needed to prepare 1L of 2.5 M solution of KCI if the available stock solution is...
A:
Q: Calculate the AH of the following reaction using the reactions given below C3HS (g) → 3 C(s) + 4 H2(...
A: The given reaction is, C3H8(g) ---> 3C(s) + 4H2(g) The equation for the given reaction are, 1) ...
Q: (d). When the bidentate neutral ethylenediamine (en, -NH2CH2CH2NH2-) reacts with Co(lII) chloride, C...
A:
Q: Solve using multisynthesis
A: The synthesis is as follows:
Q: Solve the problem: 1. The freezing pt. of a solution containing 65g of a compound in 100g benzene is...
A: With the addition of a non-volatile solute into a solvent the freezing point of the solvent will dec...
Q: Which is the best method for carrying out the following two-step transformation? CI 1. O 1. C12/FeCI...
A: The given reactant is Benzene and the product formed in the end is 3-chloro acetophenone.
Q: Arrange in terms of increasing acidic and explain these using partial charges and inductive effects
A:
Q: For every given acid or base, write the equation of their reactions with water when they donate or a...
A: Given, 1. Hydrocyanic acid 2. Carbonate ion 3. Ammonium ion 4. Sulfuric acid 5. Acetic acid The rea...
Q: Lewis reaction between Ag+ and 2 cyanide ions (CN') a. Identify the acid and base in each reaction. ...
A: According to Lewis theory of acid-base reactions, Lewis acid is a substance that accepts a pair of e...
Q: Suppose that you started with 5 ml of 200 mg/L Cu2+ solution and added it to 1.0g of soil. After mi...
A: (1) Here we are provided with 5 mL of 200 mg/L of Cu2+ solution which is added to 1.0 g of soil. Aft...
Q: Name the major organic product for the reaction of Tollen's reagent with butanal.
A:
Q: For the generic reaction below, the rate quadruples when [A] is doubled. When [B] is doubled, the ra...
A: according to the question the rate is doubled when concentration of A is doubled and remain unchange...
Q: 2 A P The rate coefficient for this elementary reaction isk = 8.63 x 10 M' min'. The initial concent...
A:

Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 3 images

- Help me please3. Rank the following in order by the rate in which they could participate in an SN1 reaction (“1" being the fastest and "5" being the slowest to react).3. Rank the following in order by the rate in which they could participate in an SN1 reaction ("“1" being the fastest and "5" being the slowest to react). CI
- 2. Which reaction will occur at a faster rate? Explain your answer. Rxn A Rxn, B Br₂ FeBr3 Br₂ FeBr39. Which SN1 reaction in each pair is faster? a) CI b) (CH3)3CCI + H2O > H2O (CH3)3CBR + H20 → CI H,O 206alcohol. A carbocation intermediate is central to which of these reaction types? (circle) E1 E2 SN1 SN2
- Rank the following from most reactive to least reactive in an SN1 reaction.Arrange the compounds below in order of increasing reactivity in a SN2 reaction. 1. (CH3)2СНCI 2. CH3CH2B1 3. CH3CH2C1 A) 1; 2; 3 В) 3;B 2; 1 C) 1; 3; 2 D) 2; 1; 3 E) 3; 1; 24. Which one of the following compounds will react faster by SN1? PhBr PhCH2Br PHCH= CHBr MezCBr Ph:CBr A E
- 5) Consider the two E2 reactions above. Br Br NaOH DMF heat NaOH DMF heat a. What are the major products for each reaction? b. What is the mechanism for each reaction? c. Which reaction would be faster and why? Use words like "transition-state, intermediate and/or reactant/product stability" in your justification. Draw the reaction coordinate diagram for both to assist in your explanation.2) Rank the following compounds in order of their reaction rates in an SN1 reaction with NaF with 1 being the fastest reaction and 5 being the slowest. OTs Br CI5. Classify the following molecules in order of decreasing rate of SN2 reactions: Ethyl Chloride, t-Butyl Chloride, Methyl Chloride and Propyl Chloride.

